Interactions between Membrane Resistance, GABA-A Receptor Properties, Bicarbonate Dynamics and Cl--Transport Shape Activity-Dependent Changes of Intracellular Cl- Concentration
- PMID: 30897846
- PMCID: PMC6471822
- DOI: 10.3390/ijms20061416
Interactions between Membrane Resistance, GABA-A Receptor Properties, Bicarbonate Dynamics and Cl--Transport Shape Activity-Dependent Changes of Intracellular Cl- Concentration
Abstract
The effects of ionotropic γ-aminobutyric acid receptor (GABA-A, GABAA) activation depends critically on the Cl--gradient across neuronal membranes. Previous studies demonstrated that the intracellular Cl--concentration ([Cl-]i) is not stable but shows a considerable amount of activity-dependent plasticity. To characterize how membrane properties and different molecules that are directly or indirectly involved in GABAergic synaptic transmission affect GABA-induced [Cl-]i changes, we performed compartmental modeling in the NEURON environment. These simulations demonstrate that GABA-induced [Cl-]i changes decrease at higher membrane resistance, revealing a sigmoidal dependency between both parameters. Increase in GABAergic conductivity enhances [Cl-]i with a logarithmic dependency, while increasing the decay time of GABAA receptors leads to a nearly linear enhancement of the [Cl-]i changes. Implementing physiological levels of HCO₃--conductivity to GABAA receptors enhances the [Cl-]i changes over a wide range of [Cl-]i, but this effect depends on the stability of the HCO₃- gradient and the intracellular pH. Finally, these simulations show that pure diffusional Cl--elimination from dendrites is slow and that a high activity of Cl--transport is required to improve the spatiotemporal restriction of GABA-induced [Cl-]i changes. In summary, these simulations revealed a complex interplay between several key factors that influence GABA-induced [Cl]i changes. The results suggest that some of these factors, including high resting [Cl-]i, high input resistance, slow decay time of GABAA receptors and dynamic HCO₃- gradient, are specifically adapted in early postnatal neurons to facilitate limited activity-dependent [Cl-]i decreases.
Keywords: CA3; Cl−-homeostasis; Na+-K+-Cl−-Cotransporter, Isoform 1 (NKCC1); computational neuroscience; development; giant depolarizing potentials; hippocampus; ionic plasticity; mouse.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


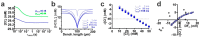
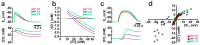
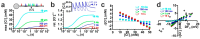
Similar articles
-
Coincident glutamatergic depolarizations enhance GABAA receptor-dependent Cl- influx in mature and suppress Cl- efflux in immature neurons.PLoS Comput Biol. 2021 Jan 19;17(1):e1008573. doi: 10.1371/journal.pcbi.1008573. eCollection 2021 Jan. PLoS Comput Biol. 2021. PMID: 33465082 Free PMC article.
-
Activity-dependent intracellular chloride accumulation and diffusion controls GABA(A) receptor-mediated synaptic transmission.Hippocampus. 2011 Aug;21(8):885-98. doi: 10.1002/hipo.20804. Epub 2010 Jun 23. Hippocampus. 2011. PMID: 20575006
-
Ectopic GABAA receptor β3 subunit determines Cl- / -ATPase and chloride transport in HEK 293FT cells.FEBS J. 2021 Jan;288(2):699-712. doi: 10.1111/febs.15359. Epub 2020 Jun 17. FEBS J. 2021. PMID: 32383536
-
Receptors for gamma-aminobutyric acid and voltage-dependent chloride channels as targets for drugs and toxicants.FASEB J. 1987 Oct;1(4):262-71. doi: 10.1096/fasebj.1.4.2443413. FASEB J. 1987. PMID: 2443413 Review.
-
Inhibitory transmission, activity-dependent ionic changes and neuronal network oscillations.Physiol Res. 2006;55(2):139-149. doi: 10.33549/physiolres.930764. Epub 2006 May 24. Physiol Res. 2006. PMID: 15910171 Review.
Cited by
-
Role of paroxysmal depolarization in focal seizure activity.J Neurophysiol. 2019 Nov 1;122(5):1861-1873. doi: 10.1152/jn.00392.2019. Epub 2019 Aug 28. J Neurophysiol. 2019. PMID: 31461373 Free PMC article. Review.
-
When Are Depolarizing GABAergic Responses Excitatory?Front Mol Neurosci. 2021 Nov 24;14:747835. doi: 10.3389/fnmol.2021.747835. eCollection 2021. Front Mol Neurosci. 2021. PMID: 34899178 Free PMC article. Review.
-
Genistein and Vanadate Differentially Modulate Cortical GABAA Receptor/ATPase Activity and Behavior in Rats via a Phenol-Sensitive Mechanism.Int J Mol Sci. 2025 Jun 15;26(12):5731. doi: 10.3390/ijms26125731. Int J Mol Sci. 2025. PMID: 40565196 Free PMC article.
-
NKCC-1 mediated Cl- uptake in immature CA3 pyramidal neurons is sufficient to compensate phasic GABAergic inputs.Sci Rep. 2020 Oct 27;10(1):18399. doi: 10.1038/s41598-020-75382-1. Sci Rep. 2020. PMID: 33110147 Free PMC article.
-
Coincident Activation of Glutamate Receptors Enhances GABAA Receptor-Induced Ionic Plasticity of the Intracellular Cl--Concentration in Dissociated Neuronal Cultures.Front Cell Neurosci. 2019 Nov 8;13:497. doi: 10.3389/fncel.2019.00497. eCollection 2019. Front Cell Neurosci. 2019. PMID: 31787883 Free PMC article.
References
-
- Farrant M., Kaila K. The cellular, molecular and ionic basis of GABA(A) receptor signaling. Prog. Brain Res. 2007;160:59–87. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

