The Clock Mechanism Influences Neurobiology and Adaptations to Heart Failure in Clock∆19/∆19 Mice With Implications for Circadian Medicine
- PMID: 30899044
- PMCID: PMC6428811
- DOI: 10.1038/s41598-019-41469-7
The Clock Mechanism Influences Neurobiology and Adaptations to Heart Failure in Clock∆19/∆19 Mice With Implications for Circadian Medicine
Abstract
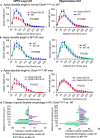
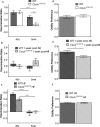
In this study we investigated the role of the circadian mechanism on cognition-relevant brain regions and neurobiological impairments associated with heart failure (HF), using murine models. We found that the circadian mechanism is an important regulator of healthy cognitive system neurobiology. Normal Clock∆19/∆19 mice had neurons with smaller apical dendrite trees in the medial prefrontal cortex (mPFC), and hippocampus, showed impaired visual-spatial memory, and exhibited lower cerebrovascular myogenic tone, versus wild types (WT). We then used the left anterior descending coronary artery ligation model to investigate adaptations in response to HF. Intriguingly, adaptations to neuron morphology, memory, and cerebrovascular tone occurred in differing magnitude and direction between Clock∆19/∆19 and WT mice, ultimately converging in HF. To investigate this dichotomous response, we performed microarrays and found genes crucial for growth and stress pathways that were altered in Clock∆19/∆19 mPFC and hippocampus. Thus these data demonstrate for the first time that (i) the circadian mechanism plays a role in neuron morphology and function; (ii) there are changes in neuron morphology and function in HF; (iii) CLOCK influences neurobiological gene adaptations to HF at a cellular level. These findings have clinical relevance as patients with HF often present with concurrent neurocognitive impairments. There is no cure for HF, and new understanding is needed to reduce morbidity and improve the quality of life for HF patients.
Conflict of interest statement
The authors declare no competing interests.
Figures




References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

