Sequential treatment of phenethyl isothiocyanate increases sensitivity of Temozolomide resistant glioblastoma cells by decreasing expression of MGMT via NF-κB pathway
- PMID: 30899372
- PMCID: PMC6413290
Sequential treatment of phenethyl isothiocyanate increases sensitivity of Temozolomide resistant glioblastoma cells by decreasing expression of MGMT via NF-κB pathway
Retraction in
-
Sequential treatment of phenethyl isothiocyanate increases sensitivity of Temozolomide resistant glioblastoma cells by decreasing expression of MGMT via NF-κB pathway [Retraction].Am J Transl Res. 2021 May 15;13(5):5746. eCollection 2021. Am J Transl Res. 2021. PMID: 34150184 Free PMC article.
Abstract
Background: Existence of acquired or intrinsic resistance to Temozolomide (TMD) remains a point of concern in treating glioblastoma (GBM). Here we established mechanism by which Phenethyl isothiocyanate (PEITC) reverses TMD resistance in T98G cell lines both in vitro and in vivo.
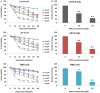
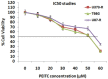
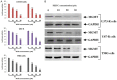
Methods: For the study TMD-resistant cell lines were generated by stepwise exposing the parental cell lines (U87 and U373) to TMD. The 50% inhibitory concentration (IC50) values were established. MTT assay was done for cell survival studies, apoptosis assay by FITC Annexin V/PI staining, luciferase reporter assay for NF-κB transcription activity, cell colony survival and cell invasion assay, protein expression by western blot was done. For in vivo studies nude mouse model of GBM was established, TUNEL assay was done for apoptosis in tumor specimens.
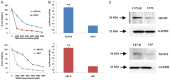
Results: We established that T98G, U87-R and U373-R showed higher NF-κB activity and exhibited higher IC50 of TMD with significantly increased MGMT expression compared to untreated cells. Next, we found that PEITC suppressed proliferation of resistant GBM cells, inhibited NF-κB activity, decreased expression of MGMT and reversed the resistance in U373-R, U87-R and T98G cells. Exposure to PEITC followed by sequential treatment of TMD produced synergistic effect. In U373-R grafted xenografts mouse model PEITC suppressed cell growth and enhanced cell death.
Conclusion: Altogether, the present research established that combination of PEITC with TMD could enhance its clinical efficacy in resistant GBM by suppressing MGMT via inhibiting NF-κB activity.
Keywords: MGMT; Temozolomide; glioblastoma; resistance.
Conflict of interest statement
None.
Figures


References
-
- Fuller GN. The WHO classification of tumours of the central nervous system. 4th edition. Arch Pathol Lab Med. 2008;132:906. - PubMed
-
- Yeom SY, Nam DH, Park C. RRAD promotes EGFRmediated STAT3 activation and induces temozolomide resistance of malignant glioblastoma. Mol Cancer Ther. 2014;13:3049–3061. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
