Evolving data analysis of an Oral Lipid Tolerance Test toward the standard for the Oral Glucose Tolerance Test: Cross species modeling effects of AZD7687 on plasma triacylglycerol
- PMID: 30899516
- PMCID: PMC6408865
- DOI: 10.1002/prp2.465
Evolving data analysis of an Oral Lipid Tolerance Test toward the standard for the Oral Glucose Tolerance Test: Cross species modeling effects of AZD7687 on plasma triacylglycerol
Abstract
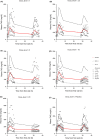
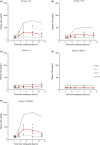
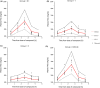
We have developed a novel mechanistic pharmacokinetic-pharmacodynamic (PK/PD) model to describe the time course of plasma triglyceride (TAG) after Oral Lipid Tolerance Test (OLTT) and the effects of AZD7687, an inhibitor of diacylglycerol acyltransferase 1 (DGAT1), in humans, rats, and mice. Pharmacokinetic and plasma TAG data were obtained both in animals and in two phase I OLTT studies. In the PK/PD model, the introduction of exogenous TAG is represented by a first order process. The endogenous production and removal of TAG from plasma are described with a turnover model. AZD7687 inhibits the contribution of exogenous TAG into circulation. One or two compartment models with first order absorption was used to describe the PK of AZD7687 for the different species. Nonlinear mixed effect modeling was used to fit the model to the data. The effects of AZD7687 on the plasma TAG time course during an OLTT as well as interindividual variability were well described by the model in all three species. Meal fat content or data from single vs repeated dosing did not affect model parameter estimates. Body mass index was found to be a significant covariate on the plasma TAG baseline. The system parameters of the model will facilitate analysis for other compounds and provide tools to bring the standard of OLTT data analysis closer to the analyses of Oral Glucose Tolerance Test data maximizing knowledge gain.
Keywords: AZD7687; DGAT1; mechanistic modeling; nonlinear mixed effect modeling; oral lipid tolerance test; pharmacodynamics; pharmacokinetics; triglyceride.
Conflict of interest statement
All authors were employees at AstraZeneca at the time the studies were carried out.
Figures

References
-
- Gabrielsson J, Hjorth S, Vogg B, et al. Modeling and design of challenge tests: inflammatory and metabolic biomarker study examples. Eur J Pharm Sci. 2015;67:144‐159. - PubMed
-
- Jauslin PM, Silber HE, Frey N, et al. An integrated glucose‐insulin model to describe oral glucose tolerance test data in type 2 diabetics. J Clin Pharmacol. 2007;47:1244‐1255. - PubMed
-
- Møller JB, Overgaard RV, Madsen H, Hansen T, Pedersen O, Ingwersen SH. Predictive performance for population models using stochastic differential equations applied on data from an oral glucose tolerance test. J Pharmacokinet Pharmacodyn. 2010;37:85‐98. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

