Diagnosis and Management of Immune Checkpoint Inhibitor-Associated Renal Toxicity: Illustrative Case and Review
- PMID: 30902916
- PMCID: PMC6656503
- DOI: 10.1634/theoncologist.2018-0764
Diagnosis and Management of Immune Checkpoint Inhibitor-Associated Renal Toxicity: Illustrative Case and Review
Abstract
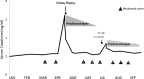
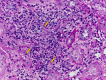
Immune checkpoint inhibitors (ICIs) are monoclonal antibodies directed at negative regulatory components on T cells, such as cytotoxic T lymphocyte-associated antigen 4, programmed cell death-1 (PD-1), and its ligand, programmed cell death ligand-1. ICIs initate antitumor immunity; however, these agents are associated with immune-related adverse events (irAEs) that may affect a variety of organs. Renal irAEs most commonly present with asymptomatic acute kidney injury (AKI), which is often detected by routine laboratory testing. The severity of AKI associated with irAEs ranges from mild (grade 1-2) to severe (grade 3-4). It is often challenging to diagnose because this group of patients often have multiple reasons to have AKI (dehydration, sepsis, or nephrotoxic medication exposure). We present an illustrative case of a 60-year-old man with metastatic melanoma who presented with AKI during treatment with nivolumab and review the literature to address frequently asked questions concerning the diagnosis and management of renal irAEs in patients with advanced cancer. Importantly, most patients will recover completely, and some may tolerate a rechallenge of ICI therapy, with prompt and effective treatment. KEY POINTS: Renal immune-related adverse events (irAEs) are less frequently reported than other irAEs; however, it is possible that available data underestimate their true incidence because of missed diagnoses and under-reporting. Although severe renal irAEs are more easily detected, smaller rises in creatinine may not be appreciated or may be attributed to other causes, because the differential diagnosis of acute kidney injury (AKI) in patients with cancer is broad.Baseline creatinine should be established prior to beginning immune checkpoint inhibitorss (ICIs), and it should be monitored with every cycle. If a patient develops AKI, the ICI should be held while the evaluation is pursued. A thorough workup of suspected renal irAEs must exclude other potential causes of AKI such as infection, dehydration, urinary tract obstruction, and nephrotoxin exposure.Acute kidney injury after ICI therapy does not appear to be more common in patients with baseline estimated glomerular filtration rate <60 mL per min per 1.73 m. One particular concern, however, is that those with baseline renal disease have less "renal reserve," and repeated AKI events may push a patient closer to end-stage renal disease. Thus, clinicians must exert caution when rechallenging patients with pre-existing renal disease with ICI therapy in the event of a prior AKI from ICI-related allergic interstitial nephritis.
© AlphaMed Press 2019.
Conflict of interest statement
Disclosures of potential conflicts of interest may be found at the end of this article.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

