Overcoming the thermodynamic equilibrium of an isomerization reaction through oxidoreductive reactions for biotransformation
- PMID: 30902987
- PMCID: PMC6430769
- DOI: 10.1038/s41467-019-09288-6
Overcoming the thermodynamic equilibrium of an isomerization reaction through oxidoreductive reactions for biotransformation
Abstract
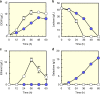
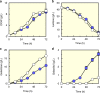
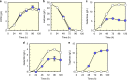
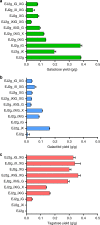
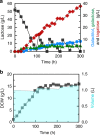
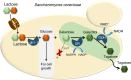
Isomerases perform biotransformations without cofactors but often cause an undesirable mixture of substrate and product due to unfavorable thermodynamic equilibria. We demonstrate the feasibility of using an engineered yeast strain harboring oxidoreductase reactions to overcome the thermodynamic limit of an isomerization reaction. Specifically, a yeast strain capable of consuming lactose intracellularly is engineered to produce tagatose from lactose through three layers of manipulations. First, GAL1 coding for galactose kinase is deleted to eliminate galactose utilization. Second, heterologous xylose reductase (XR) and galactitol dehydrogenase (GDH) are introduced into the ∆gal1 strain. Third, the expression levels of XR and GDH are adjusted to maximize tagatose production. The resulting engineered yeast produces 37.69 g/L of tagatose from lactose with a tagatose and galactose ratio of 9:1 in the reaction broth. These results suggest that in vivo oxidoreaductase reactions can be employed to replace isomerases in vitro for biotransformation.
Conflict of interest statement
J.-J.L., G.-C.Z., K.C., J.H.D.C., and Y.-S.J. are authors of a patent application filed by University of Illinois at Urbana-Champaign based on this work. K.C., J.H.D.C. and Y.-S.J. have financial interest in Sugarlogix, Inc. The remaining authors declare no competing interests.
Figures
Similar articles
-
Efficient production of d-tagatose via DNA scaffold mediated oxidoreductases assembly in vivo from whey powder.Food Res Int. 2023 Apr;166:112637. doi: 10.1016/j.foodres.2023.112637. Epub 2023 Feb 24. Food Res Int. 2023. PMID: 36914333
-
Identification of the galactitol dehydrogenase, LadB, that is part of the oxido-reductive D-galactose catabolic pathway in Aspergillus niger.Fungal Genet Biol. 2012 Feb;49(2):152-9. doi: 10.1016/j.fgb.2011.11.005. Epub 2011 Nov 30. Fungal Genet Biol. 2012. PMID: 22155165
-
Rewiring Bacillus subtilis and bioprocess optimization for oxidoreductive reaction-mediated biosynthesis of D-tagatose.Bioresour Technol. 2023 Dec;389:129843. doi: 10.1016/j.biortech.2023.129843. Epub 2023 Oct 10. Bioresour Technol. 2023. PMID: 37820967
-
Recent Advances on Biological Production of a Functional Low-Calorie Sugar d-Tagatose.J Agric Food Chem. 2025 Jul 30;73(30):18511-18524. doi: 10.1021/acs.jafc.5c04610. Epub 2025 Jul 21. J Agric Food Chem. 2025. PMID: 40690603 Review.
-
Isomerization of Galactose to Tagatose: Recent Advances in Non-enzymatic Isomerization.J Agric Food Chem. 2023 Mar 15;71(10):4228-4234. doi: 10.1021/acs.jafc.3c00095. Epub 2023 Mar 3. J Agric Food Chem. 2023. PMID: 36867179 Review.
Cited by
-
Engineering Saccharomyces cerevisiae for growth on xylose using an oxidative pathway.Appl Microbiol Biotechnol. 2025 Jan 28;109(1):30. doi: 10.1007/s00253-025-13417-1. Appl Microbiol Biotechnol. 2025. PMID: 39873813 Free PMC article.
-
A unique metabolic gene cluster regulates lactose and galactose metabolism in the yeast Candida intermedia.Appl Environ Microbiol. 2024 Oct 23;90(10):e0113524. doi: 10.1128/aem.01135-24. Epub 2024 Sep 6. Appl Environ Microbiol. 2024. PMID: 39240082 Free PMC article.
-
Stereoselective Solid-State Synthesis of Biologically Active Cyclobutane and Dicyclobutane Isomers via Conformation Blocking and Transference.Molecules. 2024 Jun 19;29(12):2909. doi: 10.3390/molecules29122909. Molecules. 2024. PMID: 38930974 Free PMC article.
-
Production of galactitol from galactose by the oleaginous yeast Rhodosporidium toruloides IFO0880.Biotechnol Biofuels. 2019 Oct 18;12:250. doi: 10.1186/s13068-019-1586-5. eCollection 2019. Biotechnol Biofuels. 2019. PMID: 31636709 Free PMC article.
-
Investigating the role of the transcriptional regulator Ure2 on the metabolism of Saccharomyces cerevisiae: a multi-omics approach.Appl Microbiol Biotechnol. 2021 Jun;105(12):5103-5112. doi: 10.1007/s00253-021-11394-9. Epub 2021 Jun 21. Appl Microbiol Biotechnol. 2021. PMID: 34152451
References
-
- Zargaraan A, Kamaliroosta L, Yaghoubi AS, Mirmoghtadaie L. Effect of substitution of sugar by high fructose corn syrup on the physicochemical properties of bakery and dairy products: a review. Nutr. Food Sci. Res. 2016;3:3–11.
-
- Shin KC, Sim DH, Seo MJ, Oh DK. Increased production of food-grade D-tagatose from D-galactose by permeabilized and immobilized cells of Corynebacterium glutamicum, a GRAS host, expressing d-galactose isomerase from Geobacillus thermodenitrificans. J. Agric. Food Chem. 2016;64:8146–8153. doi: 10.1021/acs.jafc.6b03588. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

