The synergistic effects of clopidogrel with montelukast may be beneficial for asthma treatment
- PMID: 30905080
- PMCID: PMC6484307
- DOI: 10.1111/jcmm.14239
The synergistic effects of clopidogrel with montelukast may be beneficial for asthma treatment
Abstract
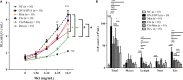
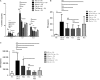
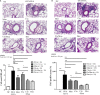
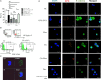
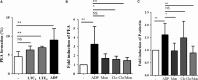
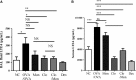
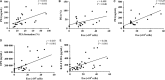
Platelets modulate asthma pathogenesis by forming the platelet-eosinophil aggregation (PEA), which facilitates the activation of eosinophils. Platelets exhibit the purinergic receptor (P2Y12R), which responds to cysteinyl leukotriene E4 (LTE4 ). We have suggested that the combination of an antiplatelet drug (clopidogrel, [Clo]) and montelukast (Mon) would synergistically suppress asthma. BALB/c mice were intraperitoneally sensitized with ovalbumin (OVA) on days 0 and 14 and subsequently challenged on days 28-30 and 42-44. Mice were administered with Clo (10 mg/kg), Mon (10 mg/kg) or both drugs (Clo/Mon) orally 30 minutes before the OVA (1%) challenge on days 42-44. Mice were assayed for airway hyper-responsiveness (AHR) to methacholine and airway inflammation. Clopidogrel and montelukast attenuated the increased AHR; the combined treatment was more effective than a single treatment for total and eosinophil counts (all P < 0.05). Levels of interleukin (IL)-4, IL-5, IL-13, platelet factor 4, eosinophil peroxidase and LTE4 increased in the bronchoalveolar lavage fluid of asthmatic mice, but these levels decreased in mice treated with Clo/Mon (all P < 0.05). Goblet cell hyperplasia decreased in response to Clo/Mon. Mouse platelets and eosinophils were isolated and co-cultured for an in vitro assay with 10 µmol/L adenosine diphosphate (ADP), LTE4 (200 nmol/L), Mon (1 µmol/L), Clo (1 µmol/L) and Clo/Mon (1 µmol/L). Flow cytometry revealed that the increased formation of the PEA (%) was fully mediated by ADP and partly mediated by LTE4 . Clo/Mon reduced ADP-induced PEA formation and P-selectin expression (P < 0.05). In conclusion, Clo/Mon synergistically relieved asthma by inhibiting ADP-mediated PEA formation.
Keywords: P2Y12R; asthma; clopidogrel; cysteinyl leukotriene receptors; eosinophil; montelukast; rodent.
© 2019 The Authors. Journal of Cellular and Molecular Medicine published by John Wiley & Sons Ltd and Foundation for Cellular and Molecular Medicine.
Conflict of interest statement
The authors confirm that there is no conflict of interest.
Figures
Similar articles
-
P2Y12 antagonist attenuates eosinophilic inflammation and airway hyperresponsiveness in a mouse model of asthma.J Cell Mol Med. 2016 Feb;20(2):333-41. doi: 10.1111/jcmm.12727. Epub 2015 Nov 27. J Cell Mol Med. 2016. PMID: 26612496 Free PMC article.
-
Characterization of cysteinyl leukotriene-related receptors and their interactions in a mouse model of asthma.Prostaglandins Leukot Essent Fatty Acids. 2019 Feb;141:17-23. doi: 10.1016/j.plefa.2018.12.002. Epub 2018 Dec 12. Prostaglandins Leukot Essent Fatty Acids. 2019. PMID: 30661601
-
The effects of low dose leukotriene receptor antagonist therapy on airway remodeling and cysteinyl leukotriene expression in a mouse asthma model.Exp Mol Med. 2006 Apr 30;38(2):109-18. doi: 10.1038/emm.2006.14. Exp Mol Med. 2006. PMID: 16672764
-
Subsets of Eosinophils in Asthma, a Challenge for Precise Treatment.Int J Mol Sci. 2023 Mar 16;24(6):5716. doi: 10.3390/ijms24065716. Int J Mol Sci. 2023. PMID: 36982789 Free PMC article. Review.
-
[Possible therapeutic use of L-type amino acid transporter 1 (LAT1)-specific inhibitor for intractable asthma treatment].Nihon Yakurigaku Zasshi. 2022;157(5):305-309. doi: 10.1254/fpj.22020. Nihon Yakurigaku Zasshi. 2022. PMID: 36047140 Review. Japanese.
Cited by
-
Th2 single-cell heterogeneity and clonal distribution at distant sites in helminth-infected mice.Elife. 2022 Aug 11;11:e74183. doi: 10.7554/eLife.74183. Elife. 2022. PMID: 35950748 Free PMC article.
-
Role and recent progress of P2Y12 receptor in cancer development.Purinergic Signal. 2024 Jun 14. doi: 10.1007/s11302-024-10027-w. Online ahead of print. Purinergic Signal. 2024. PMID: 38874752 Review.
-
The Signaling Pathway of the ADP Receptor P2Y12 in the Immune System: Recent Discoveries and New Challenges.Int J Mol Sci. 2023 Apr 4;24(7):6709. doi: 10.3390/ijms24076709. Int J Mol Sci. 2023. PMID: 37047682 Free PMC article. Review.
-
Platelets, Not an Insignificant Player in Development of Allergic Asthma.Cells. 2021 Aug 10;10(8):2038. doi: 10.3390/cells10082038. Cells. 2021. PMID: 34440807 Free PMC article. Review.
-
Extracellular Adenosine Diphosphate Stimulates CXCL10-Mediated Mast Cell Infiltration Through P2Y1 Receptor to Aggravate Airway Inflammation in Asthmatic Mice.Front Mol Biosci. 2021 Jul 5;8:621963. doi: 10.3389/fmolb.2021.621963. eCollection 2021. Front Mol Biosci. 2021. PMID: 34291079 Free PMC article.
References
-
- Takeda T, Morita H, Saito H, Matsumoto K, Matsuda A. Recent advances in understanding the roles of blood platelets in the pathogenesis of allergic inflammation and bronchial asthma. Allergol Int. 2017;67:326‐333. - PubMed
-
- Idzko M, Pitchford S, Page C. Role of platelets in allergic airway inflammation. J Allergy Clin Immunol. 2015;135:1416‐1423. - PubMed
-
- von Hundelshausen P, Weber C. Platelets as immune cells: bridging inflammation and cardiovascular disease. Circ Res. 2007;100:27‐40. - PubMed
-
- Semple JW, Italiano JE, Freedman J. Platelets and the immune continuum. Nat Rev Immunol. 2011;11:264. - PubMed
-
- Diacovo TG, Puri KD, Warnock RA, Springer TA, von Andrian UH. Platelet‐mediated lymphocyte delivery to high endothelial venules. Science. 1996;273:252‐255. - PubMed

