CD8+ cytotoxic T cell responses to dominant tumor-associated antigens are profoundly weakened by aging yet subdominant responses retain functionality and expand in response to chemotherapy
- PMID: 30906657
- PMCID: PMC6422383
- DOI: 10.1080/2162402X.2018.1564452
CD8+ cytotoxic T cell responses to dominant tumor-associated antigens are profoundly weakened by aging yet subdominant responses retain functionality and expand in response to chemotherapy
Abstract
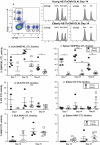
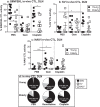
Increasing life expectancy is associated with increased cancer incidence, yet the effect of cancer and anti-cancer treatment on elderly patients and their immune systems is not well understood. Declining T cell function with aging in response to infection and vaccination is well documented, however little is known about aged T cell responses to tumor antigens during cancer progression or how these responses are modulated by standard chemotherapy. We examined T cell responses to cancer in aged mice using AE17sOVA mesothelioma in which ovalbumin (OVA) becomes a 'spy' tumor antigen containing one dominant (SIINFEKL) and two subdominant (KVVRFDKL and NAIVFKGL) epitopes. Faster progressing tumors in elderly (22-24 months, cf. 60-70 human years) relative to young (2-3 months, human 15-18 years) mice were associated with increased pro-inflammatory cytokines and worsened cancer cachexia. Pentamer staining and an in-vivo cytotoxic T lymphocyte (CTL) assay showed that whilst elderly mice generated a greater number of CD8+ T cells recognizing all epitopes, they exhibited a profound loss of function in their ability to lyse targets expressing the dominant, but not subdominant, epitopes compared to young mice. Chemotherapy was less effective and more toxic in elderly mice however, similar to young mice, chemotherapy expanded CTLs recognizing at least one subdominant epitope in tumors and draining lymph nodes, yet treatment efficacy still required CD8+ T cells. Given the significant dysfunction associated with elderly CTLs recognizing dominant epitopes, our data suggest that responses to subdominant tumor epitopes may become important when elderly hosts with cancer are treated with chemotherapy.
Keywords: Solid tumors; dominant and subdominant CD8+ T cell responses; elderly hosts with cancer; hierarchical T cell responses; mesothelioma; standard chemotherapy.
Figures






Similar articles
-
The STING agonist, DMXAA, reduces tumor vessels and enhances mesothelioma tumor antigen presentation yet blunts cytotoxic T cell function in a murine model.Front Immunol. 2022 Nov 18;13:969678. doi: 10.3389/fimmu.2022.969678. eCollection 2022. Front Immunol. 2022. PMID: 36466911 Free PMC article.
-
Chemotherapy broadens the range of tumor antigens seen by cytotoxic CD8(+) T cells in vivo.Cancer Immunol Immunother. 2012 Dec;61(12):2343-56. doi: 10.1007/s00262-012-1307-4. Epub 2012 Jun 20. Cancer Immunol Immunother. 2012. PMID: 22714286 Free PMC article.
-
In vivo cross-presentation of a soluble protein antigen: kinetics, distribution, and generation of effector CTL recognizing dominant and subdominant epitopes.J Immunol. 2000 Dec 1;165(11):6123-32. doi: 10.4049/jimmunol.165.11.6123. J Immunol. 2000. PMID: 11086045
-
Inefficient cross-presentation limits the CD8+ T cell response to a subdominant tumor antigen epitope.J Immunol. 2005 Jul 15;175(2):700-12. doi: 10.4049/jimmunol.175.2.700. J Immunol. 2005. PMID: 16002665
-
Revealing factors determining immunodominant responses against dominant epitopes.Immunogenetics. 2020 Feb;72(1-2):109-118. doi: 10.1007/s00251-019-01134-9. Epub 2019 Dec 6. Immunogenetics. 2020. PMID: 31811313 Free PMC article. Review.
Cited by
-
The effects of age and systemic metabolism on anti-tumor T cell responses.Elife. 2020 Nov 10;9:e62420. doi: 10.7554/eLife.62420. Elife. 2020. PMID: 33170123 Free PMC article. Review.
-
Immunosenescence and cancer: molecular hallmarks, tumor microenvironment remodeling, and age-specific immunotherapy challenges.J Hematol Oncol. 2025 Aug 22;18(1):81. doi: 10.1186/s13045-025-01735-w. J Hematol Oncol. 2025. PMID: 40846970 Free PMC article. Review.
-
Using a Prime-Boost Vaccination Strategy That Proved Effective for High Resolution Epitope Mapping to Characterize the Elusive Immunogenicity of Survivin.Cancers (Basel). 2021 Dec 14;13(24):6270. doi: 10.3390/cancers13246270. Cancers (Basel). 2021. PMID: 34944889 Free PMC article.
-
Vesicular Stomatitis Virus Encoding a Destabilized Tumor Antigen Improves Activation of Anti-tumor T Cell Responses.Mol Ther. 2020 Dec 2;28(12):2540-2552. doi: 10.1016/j.ymthe.2020.08.013. Epub 2020 Aug 25. Mol Ther. 2020. PMID: 32877695 Free PMC article.
-
The STING agonist, DMXAA, reduces tumor vessels and enhances mesothelioma tumor antigen presentation yet blunts cytotoxic T cell function in a murine model.Front Immunol. 2022 Nov 18;13:969678. doi: 10.3389/fimmu.2022.969678. eCollection 2022. Front Immunol. 2022. PMID: 36466911 Free PMC article.
References
-
- United Nations World population prospects. 2017. [accessed 2017 Oct 19]. http://www.pewglobal.org/2014/01/30/global-population/.
-
- Odgerel CO, Takahashi K, Sorahan T, Driscoll T, Fitzmaurice C, Yoko OM, Sawanyawisuth K, Furuya S, Tanaka F, Horie S, et al. Estimation of the global burden of mesothelioma deaths from incomplete national mortality data. Occup Environ Med. 2017;74:851–858. doi:10.1136/oemed-2017-104298. - DOI - PMC - PubMed
-
- Bianchi C, Bianchi T.. Malignant mesothelioma: global incidence and relationship with asbestos. Ind Health. 2007;45:379–387. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
