Targeting senescent cells alleviates obesity-induced metabolic dysfunction
- PMID: 30907060
- PMCID: PMC6516193
- DOI: 10.1111/acel.12950
Targeting senescent cells alleviates obesity-induced metabolic dysfunction
Abstract
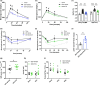
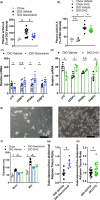
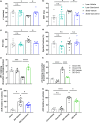
Adipose tissue inflammation and dysfunction are associated with obesity-related insulin resistance and diabetes, but mechanisms underlying this relationship are unclear. Although senescent cells accumulate in adipose tissue of obese humans and rodents, a direct pathogenic role for these cells in the development of diabetes remains to be demonstrated. Here, we show that reducing senescent cell burden in obese mice, either by activating drug-inducible "suicide" genes driven by the p16Ink4a promoter or by treatment with senolytic agents, alleviates metabolic and adipose tissue dysfunction. These senolytic interventions improved glucose tolerance, enhanced insulin sensitivity, lowered circulating inflammatory mediators, and promoted adipogenesis in obese mice. Elimination of senescent cells also prevented the migration of transplanted monocytes into intra-abdominal adipose tissue and reduced the number of macrophages in this tissue. In addition, microalbuminuria, renal podocyte function, and cardiac diastolic function improved with senolytic therapy. Our results implicate cellular senescence as a causal factor in obesity-related inflammation and metabolic derangements and show that emerging senolytic agents hold promise for treating obesity-related metabolic dysfunction and its complications.
Keywords: adipogenesis; aging; cellular senescence; dasatinib; quercetin; senolytics; type 2 diabetes.
© 2019 The Authors. Aging Cell published by the Anatomical Society and John Wiley & Sons Ltd.
Conflict of interest statement
J.L.K., T.T., T.P., A.K.P., Y.Z., M.X., J.C., and M.D. have a financial interest related to this research. Patents on senolytic drugs are held by Mayo Clinic. This research has been reviewed by the Mayo Clinic and Buck Institute Conflict of Interest Review Boards and was conducted in compliance with Mayo Clinic and Buck Institute Conflict of Interest policies. None of the other authors has a relevant conflict of financial interest.
Figures


References
-
- Christopher, L. J. , Cui, D. , Wu, C. , Luo, R. , Manning, J. A. , Bonacorsi, S. J. , … Iyer, R. A. (2008). Metabolism and disposition of dasatinib after oral administration to humans. Drug Metabolism and Disposition: the Biological Fate of Chemicals, 36, 1357–1364. 10.1124/dmd.107.018267 - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 AG053832/AG/NIA NIH HHS/United States
- BB/K019260/1/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- F30 AG046061/AG/NIA NIH HHS/United States
- R01 CA233790/CA/NCI NIH HHS/United States
- 24009/CRUK_/Cancer Research UK/United Kingdom
- BB/S006710/1/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- R01 AG013925/AG/NIA NIH HHS/United States
- R24 AG044396/AG/NIA NIH HHS/United States
- P01 AG031736/AG/NIA NIH HHS/United States
- K23 DK109134/DK/NIDDK NIH HHS/United States
- R01 HL141819/HL/NHLBI NIH HHS/United States
- P30 DK050456/DK/NIDDK NIH HHS/United States
- R37 AG013925/AG/NIA NIH HHS/United States
- P30 CA016058/CA/NCI NIH HHS/United States
- P01 AG041122/AG/NIA NIH HHS/United States
- R01 AG058812/AG/NIA NIH HHS/United States
- BB/I020748/1/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- BB/M023389/1/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- T32 GM065841/GM/NIGMS NIH HHS/United States
- K99 AG051661/AG/NIA NIH HHS/United States
- R00 AG051661/AG/NIA NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

