Antifungal and plant growth promotion activity of volatile organic compounds produced by Bacillus amyloliquefaciens
- PMID: 30907064
- PMCID: PMC6692555
- DOI: 10.1002/mbo3.813
Antifungal and plant growth promotion activity of volatile organic compounds produced by Bacillus amyloliquefaciens
Abstract
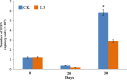
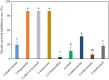
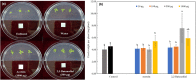
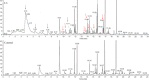
Fusarium wilt of watermelon, caused by F. oxysporum f.sp. niveum (FON), is a devastating disease that causes extensive losses throughout the world. Five bacterial strains (L3, h, β, b, and L) isolated from the watermelon rhizosphere showed antagonistic activity against FON during in vitro tests. Strain L3 produced diffusible and volatile organic compounds (VOCs) which showed the strongest antifungal activity. Arabidopsis thaliana plantlets exposed to VOCs produced by strain L3 showed a 2.39-fold increase in biomass, 1.40-fold increase in primary root length, and 5.05-fold increase in number of lateral roots. Confocal laser scanning microscope showed that the GFP-labeled strain L3 could colonize along the elongation and differentiation zones of watermelon roots. In greenhouse pot experiments, the biocontrol efficiency of strain L3 against fusarium wilt of watermelon was up to 68.4% in comparison with the control treatment. In addition, inoculation of the strain L3 resulted in a 23.4% increase in plant fresh weight. Based on 16S rDNA sequence analysis, the strain L3 was identified as Bacillus amyloliquefaciens L3. Fourteen VOCs produced by strain L3 were identified through GC-MS analysis. Of nine VOCs tested, 2-nonanone and 2-heptanone were proved to have strong antifungal properties. Acetoin and 2,3-butanediol were found to promote plant growth. The results suggested B. amyloliquefaciens L3 was a potential biocontrol agent, and that VOCs produced by B. amyloliquefaciens L3 play important roles in the process of biocontrol and plant growth promotion.
Keywords: Bacillus amyloliquefaciens; F. oxysporum f.sp. niveum; root colonization; volatile organic compounds; watermelon.
© 2019 The Authors. MicrobiologyOpen published by John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare that there is no conflict of interests.
Figures





Similar articles
-
Components of rhizospheric bacterial communities of barley and their potential for plant growth promotion and biocontrol of Fusarium wilt of watermelon.Braz J Microbiol. 2019 Jul;50(3):749-757. doi: 10.1007/s42770-019-00089-z. Epub 2019 May 20. Braz J Microbiol. 2019. PMID: 31111431 Free PMC article.
-
Suppression of Fusarium Wilt in Watermelon by Bacillus amyloliquefaciens DHA55 through Extracellular Production of Antifungal Lipopeptides.J Fungi (Basel). 2023 Mar 9;9(3):336. doi: 10.3390/jof9030336. J Fungi (Basel). 2023. PMID: 36983504 Free PMC article.
-
Bacillus amyloliquefaciens subsp. plantarum GR53, a potent biocontrol agent resists Rhizoctonia disease on Chinese cabbage through hormonal and antioxidants regulation.World J Microbiol Biotechnol. 2015 Oct;31(10):1517-27. doi: 10.1007/s11274-015-1896-0. Epub 2015 Jul 10. World J Microbiol Biotechnol. 2015. PMID: 26160009
-
Biocontrol Ability of the Bacillus amyloliquefaciens Group, B. amyloliquefaciens, B. velezensis, B. nakamurai, and B. siamensis, for the Management of Fungal Postharvest Diseases: A Review.J Agric Food Chem. 2022 Jun 8;70(22):6591-6616. doi: 10.1021/acs.jafc.2c01745. Epub 2022 May 23. J Agric Food Chem. 2022. PMID: 35604328 Review.
-
Bacillus amyloliquefaciens as an excellent agent for biofertilizer and biocontrol in agriculture: An overview for its mechanisms.Microbiol Res. 2022 Jun;259:127016. doi: 10.1016/j.micres.2022.127016. Epub 2022 Mar 29. Microbiol Res. 2022. PMID: 35390741 Review.
Cited by
-
Microbial Consortia for Plant Protection against Diseases: More than the Sum of Its Parts.Int J Mol Sci. 2023 Jul 31;24(15):12227. doi: 10.3390/ijms241512227. Int J Mol Sci. 2023. PMID: 37569603 Free PMC article. Review.
-
Isolation and identification of antagonistic Bacillus amyloliquefaciens HSE-12 and its effects on peanut growth and rhizosphere microbial community.Front Microbiol. 2023 Oct 12;14:1274346. doi: 10.3389/fmicb.2023.1274346. eCollection 2023. Front Microbiol. 2023. PMID: 37901825 Free PMC article.
-
Bactericidal Effect of Pseudomonas oryziphila sp. nov., a Novel Pseudomonas Species Against Xanthomonas oryzae Reduces Disease Severity of Bacterial Leaf Streak of Rice.Front Microbiol. 2021 Nov 4;12:759536. doi: 10.3389/fmicb.2021.759536. eCollection 2021. Front Microbiol. 2021. PMID: 34803984 Free PMC article.
-
Biocontrol Potential of a Novel Endophytic Bacterium From Mulberry (Morus) Tree.Front Bioeng Biotechnol. 2020 Jan 23;7:488. doi: 10.3389/fbioe.2019.00488. eCollection 2019. Front Bioeng Biotechnol. 2020. PMID: 32039187 Free PMC article.
-
Characterization of Bacillus amyloliquefaciens BA-4 and its biocontrol potential against Fusarium-related apple replant disease.Front Plant Sci. 2024 Apr 19;15:1370440. doi: 10.3389/fpls.2024.1370440. eCollection 2024. Front Plant Sci. 2024. PMID: 38708392 Free PMC article.
References
-
- Cao, Y. , Zhang, Z. H. , & Ling, N. (2011). Bacillus subtilis SQR9 can control Fusarium wilt in cucumber by colonizing plant roots. Biology and Fertility of Soils, 47, 495–506. 10.1007/s00374-011-0556-2 - DOI
-
- Chang, T. H. , Ling, Y. H. , Chen, K. S. , & Huang, J. W. (2015). Cell wall reinforcement in watermelon shot base related to its resistance to Fusarium wilt caused by Fusarium oxysporum f.sp. niveum . The Journal of Agricultural Science, 153, 296–305.
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources
Miscellaneous

