High Rate of Serotype V Streptococcus agalactiae Carriage in Pregnant Women in Botswana
- PMID: 30915949
- PMCID: PMC6493924
- DOI: 10.4269/ajtmh.18-0847
High Rate of Serotype V Streptococcus agalactiae Carriage in Pregnant Women in Botswana
Abstract
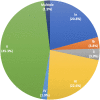
Maternal rectovaginal colonization is the major risk factor for early-onset neonatal sepsis due to Group B Streptococcus (GBS), a major cause of early life morbidity and mortality. Transmission generally occurs perinatally from colonized mothers to infants. Vaccines targeting a subset of GBS serotypes are under development, but GBS epidemiology remains poorly understood in many African nations. We performed a cross-sectional study of GBS colonization among pregnant women at two sites in Botswana, a country with minimal prior GBS carriage data. We found a rectovaginal colonization rate of 19%, comparable with studies in other regions; however, we also noted a striking predominance of serotype V (> 45% of strains). Although further studies are required to delineate the burden of invasive GBS disease in Botswana and the generalizability of type V epidemiology, these data provide a useful baseline for understanding the potential local impact of GBS prevention strategies, including vaccines.
Conflict of interest statement
Disclosure: A. J. R. receives consulting fees from Pfizer.
Figures
References
-
- Verani JR, McGee L, Schrag SJ, 2010. Prevention of perinatal group B streptococcal disease—revised guidelines from CDC, 2010. MMWR Recomm Rep 59: 1–36. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical