The Role of Tyrosine Phosphorylation of Protein Kinase C Delta in Infection and Inflammation
- PMID: 30917487
- PMCID: PMC6471617
- DOI: 10.3390/ijms20061498
The Role of Tyrosine Phosphorylation of Protein Kinase C Delta in Infection and Inflammation
Abstract
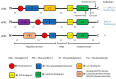
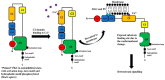
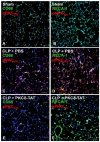
Protein Kinase C (PKC) is a family composed of phospholipid-dependent serine/threonine kinases that are master regulators of inflammatory signaling. The activity of different PKCs is context-sensitive and these kinases can be positive or negative regulators of signaling pathways. The delta isoform (PKCδ) is a critical regulator of the inflammatory response in cancer, diabetes, ischemic heart disease, and neurodegenerative diseases. Recent studies implicate PKCδ as an important regulator of the inflammatory response in sepsis. PKCδ, unlike other members of the PKC family, is unique in its regulation by tyrosine phosphorylation, activation mechanisms, and multiple subcellular targets. Inhibition of PKCδ may offer a unique therapeutic approach in sepsis by targeting neutrophil-endothelial cell interactions. In this review, we will describe the overall structure and function of PKCs, with a focus on the specific phosphorylation sites of PKCδ that determine its critical role in cell signaling in inflammatory diseases such as sepsis. Current genetic and pharmacological tools, as well as in vivo models, that are used to examine the role of PKCδ in inflammation and sepsis are presented and the current state of emerging tools such as microfluidic assays in these studies is described.
Keywords: PKC; PKCδ; inflammation; microfluidics; phosphorylation; sepsis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Inoue M., Kishimoto A., Takai Y., Nishizuka Y. Studies on a cyclic nucleotide-independent protein kinase and its proenzyme in mammalian tissues. II. Proenzyme and its activation by calcium-dependent protease from rat brain. J. Biol. Chem. 1977;252:7610–7616. - PubMed
-
- Sabri A., Steinberg S.F. Biochemistry of Hypertrophy and Heart Failure. Springer; New York, NY, USA: 2003. Protein kinase C isoform-selective signals that lead to cardiac hypertrophy and the progression of heart failure; pp. 97–101. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

