Serine proteases as luminal mediators of intestinal barrier dysfunction and symptom severity in IBS
- PMID: 30923071
- PMCID: PMC6765451
- DOI: 10.1136/gutjnl-2018-317416
Serine proteases as luminal mediators of intestinal barrier dysfunction and symptom severity in IBS
Abstract
Objective: The intestinal lumen contains several proteases. Our aim was to determine the role of faecal proteases in mediating barrier dysfunction and symptoms in IBS.
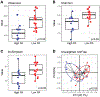
Design: 39 patients with IBS and 25 healthy volunteers completed questionnaires, assessments of in vivo permeability, ex vivo colonic barrier function in Ussing chambers, tight junction (TJ) proteins, ultrastructural morphology and 16 s sequencing of faecal microbiota rRNA. A casein-based assay was used to measure proteolytic activity (PA) in faecal supernatants (FSNs). Colonic barrier function was determined in mice (ex-germ free) humanised with microbial communities associated with different human PA states.
Results: Patients with IBS had higher faecal PA than healthy volunteers. 8/20 postinfection IBS (PI-IBS) and 3/19 constipation- predominant IBS had high PA (>95th percentile). High-PA patients had more and looser bowel movements, greater symptom severity and higher in vivo and ex vivo colonic permeability. High-PA FSNs increased paracellular permeability, decreased occludin and increased phosphorylated myosin light chain (pMLC) expression. Serine but not cysteine protease inhibitor significantly blocked high-PA FSN effects on barrier. The effects on barrier were diminished by pharmacological or siRNA inhibition of protease activated receptor-2 (PAR-2). Patients with high-PA IBS had lower occludin expression, wider TJs on biopsies and reduced microbial diversity than patients with low PA. Mice humanised with high-PA IBS microbiota had greater in vivo permeability than those with low-PA microbiota.
Conclusion: A subset of patients with IBS, especially in PI-IBS, has substantially high faecal PA, greater symptoms, impaired barrier and reduced microbial diversity. Commensal microbiota affects luminal PA that can influence host barrier function.
Keywords: Campylobacter; gastroenteritis; germ-free mice; microbiome; trypsin.
© Author(s) (or their employer(s)) 2020. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: MG has served on the advisory board or received research support from Takeda, DongA, Ironwood and Napo.
Figures





References
-
- Biancheri P, Di Sabatino A, Corazza GR, et al. Proteases and the gut barrier. Cell Tissue Res 2013;351(2):269–80. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
