(Pro)renin receptor knockdown in the paraventricular nucleus of the hypothalamus attenuates hypertension development and AT1 receptor-mediated calcium events
- PMID: 30925093
- PMCID: PMC6620680
- DOI: 10.1152/ajpheart.00780.2018
(Pro)renin receptor knockdown in the paraventricular nucleus of the hypothalamus attenuates hypertension development and AT1 receptor-mediated calcium events
Abstract
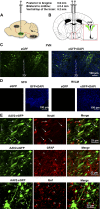
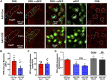
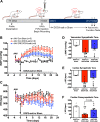
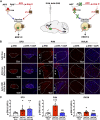
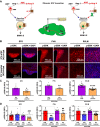
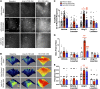
Activation of the brain renin-angiotensin system (RAS) is a pivotal step in the pathogenesis of hypertension. The paraventricular nucleus (PVN) of the hypothalamus is a critical part of the angiotensinergic sympatho-excitatory neuronal network involved in neural control of blood pressure and hypertension. However, the importance of the PVN (pro)renin receptor (PVN-PRR)-a key component of the brain RAS-in hypertension development has not been examined. In this study, we investigated the involvement and mechanisms of the PVN-PRR in DOCA-salt-induced hypertension, a mouse model of hypertension. Using nanoinjection of adeno-associated virus-mediated Cre recombinase expression to knock down the PRR specifically in the PVN, we report here that PVN-PRR knockdown attenuated the enhanced blood pressure and sympathetic tone associated with hypertension. Mechanistically, we found that PVN-PRR knockdown was associated with reduced activation of ERK (extracellular signal-regulated kinase)-1/2 in the PVN and rostral ventrolateral medulla during hypertension. In addition, using the genetically encoded Ca2+ biosensor GCaMP6 to monitor Ca2+-signaling events in the neurons of PVN brain slices, we identified a reduction in angiotensin II type 1 receptor-mediated Ca2+ activity as part of the mechanism by which PVN-PRR knockdown attenuates hypertension. Our study demonstrates an essential role of the PRR in PVN neurons in hypertension through regulation of ERK1/2 activation and angiotensin II type 1 receptor-mediated Ca2+ activity. NEW & NOTEWORTHY PRR knockdown in PVN neurons attenuates the development of DOCA-salt hypertension and autonomic dysfunction through a decrease in ERK1/2 activation in the PVN and RVLM during hypertension. In addition, PRR knockdown reduced AT1aR expression and AT1R-mediated calcium activity during hypertension. Furthermore, we characterized the neuronal targeting specificity of AAV serotype 2 in the mouse PVN and validated the advantages of the genetically encoded calcium biosensor GCaMP6 in visualizing neuronal calcium activity in the PVN.
Keywords: (pro)renin receptor; GCaMP6; calcium; central nervous system; hypertension; paraventricular nucleus of the hypothalamus; renin-angiotensin system.
Conflict of interest statement
No conflicts of interest, financial or otherwise, are declared by the authors.
Figures


Similar articles
-
Angiotensin II regulates brain (pro)renin receptor expression through activation of cAMP response element-binding protein.Am J Physiol Regul Integr Comp Physiol. 2015 Jul 15;309(2):R138-47. doi: 10.1152/ajpregu.00319.2014. Epub 2015 May 20. Am J Physiol Regul Integr Comp Physiol. 2015. PMID: 25994957 Free PMC article.
-
Intracerebroventricular infusion of the (Pro)renin receptor antagonist PRO20 attenuates deoxycorticosterone acetate-salt-induced hypertension.Hypertension. 2015 Feb;65(2):352-61. doi: 10.1161/HYPERTENSIONAHA.114.04458. Epub 2014 Nov 24. Hypertension. 2015. PMID: 25421983 Free PMC article.
-
Overexpression of the neuronal human (pro)renin receptor mediates angiotensin II-independent blood pressure regulation in the central nervous system.Am J Physiol Heart Circ Physiol. 2018 Mar 1;314(3):H580-H592. doi: 10.1152/ajpheart.00310.2017. Epub 2017 Dec 15. Am J Physiol Heart Circ Physiol. 2018. PMID: 29350998 Free PMC article.
-
Mechanisms of renal sympathetic activation in renovascular hypertension.Exp Physiol. 2015 Apr 20;100(5):496-501. doi: 10.1113/expphysiol.2014.079855. Exp Physiol. 2015. PMID: 25639235 Review.
-
Regulation of sympathetic vasomotor activity by the hypothalamic paraventricular nucleus in normotensive and hypertensive states.Am J Physiol Heart Circ Physiol. 2018 Nov 1;315(5):H1200-H1214. doi: 10.1152/ajpheart.00216.2018. Epub 2018 Aug 10. Am J Physiol Heart Circ Physiol. 2018. PMID: 30095973 Free PMC article. Review.
Cited by
-
The hypothalamus as a key regulator of glucose homeostasis: emerging roles of the brain renin-angiotensin system.Am J Physiol Cell Physiol. 2023 Jul 1;325(1):C141-C154. doi: 10.1152/ajpcell.00533.2022. Epub 2023 Jun 5. Am J Physiol Cell Physiol. 2023. PMID: 37273237 Free PMC article. Review.
-
Metabolic and behavioral alterations associated with viral vector-mediated toxicity in the paraventricular hypothalamic nucleus.Biosci Rep. 2024 Jan 16;44(1):BSR20231846. doi: 10.1042/BSR20231846. Online ahead of print. Biosci Rep. 2024. PMID: 38227343 Free PMC article.
-
The Renin-Angiotensin System in the Central Nervous System and Its Role in Blood Pressure Regulation.Curr Hypertens Rep. 2020 Jan 10;22(1):7. doi: 10.1007/s11906-019-1011-2. Curr Hypertens Rep. 2020. PMID: 31925571 Free PMC article. Review.
-
Capsaicin pretreatment attenuates salt-sensitive hypertension by alleviating AMPK/Akt/Nrf2 pathway in hypothalamic paraventricular nucleus.Front Neurosci. 2024 May 30;18:1416522. doi: 10.3389/fnins.2024.1416522. eCollection 2024. Front Neurosci. 2024. PMID: 38872941 Free PMC article.
-
Inhibition of CB1R in the Hypothalamic Paraventricular Nucleus Ameliorates Hypertension Through Wnt/β-Catenin/RAS Pathway.Cardiovasc Toxicol. 2025 Jan;25(1):9-23. doi: 10.1007/s12012-024-09938-2. Epub 2024 Oct 28. Cardiovasc Toxicol. 2025. PMID: 39467886
References
-
- Blaustein MP, Leenen FHH, Chen L, Golovina VA, Hamlyn JM, Pallone TL, Van Huysse JW, Zhang J, Wier WG. How NaCl raises blood pressure: a new paradigm for the pathogenesis of salt-dependent hypertension. Am J Physiol Heart Circ Physiol 302: H1031–H1049, 2012. doi:10.1152/ajpheart.00899.2011. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

