Telomere Dysfunction Induces Sirtuin Repression that Drives Telomere-Dependent Disease
- PMID: 30930169
- PMCID: PMC6657508
- DOI: 10.1016/j.cmet.2019.03.001
Telomere Dysfunction Induces Sirtuin Repression that Drives Telomere-Dependent Disease
Abstract
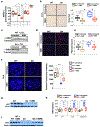
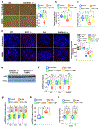
Telomere shortening is associated with stem cell decline, fibrotic disorders, and premature aging through mechanisms that are incompletely understood. Here, we show that telomere shortening in livers of telomerase knockout mice leads to a p53-dependent repression of all seven sirtuins. P53 regulates non-mitochondrial sirtuins (Sirt1, 2, 6, and 7) post-transcriptionally through microRNAs (miR-34a, 26a, and 145), while the mitochondrial sirtuins (Sirt3, 4, and 5) are regulated in a peroxisome proliferator-activated receptor gamma co-activator 1 alpha-/beta-dependent manner at the transcriptional level. Administration of the NAD(+) precursor nicotinamide mononucleotide maintains telomere length, dampens the DNA damage response and p53, improves mitochondrial function, and, functionally, rescues liver fibrosis in a partially Sirt1-dependent manner. These studies establish sirtuins as downstream targets of dysfunctional telomeres and suggest that increasing Sirt1 activity alone or in combination with other sirtuins stabilizes telomeres and mitigates telomere-dependent disorders.
Keywords: liver disease; metabolism; p53; sirtuins; telomeres.
Copyright © 2019 Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of Interests
J.A.B. is an inventor on a patent involving the use of NAD precursors to treat liver injuries. D.A.S. is a founder, equity owner, board member, advisor to, director of, consultant to, investor in and/or inventor on patents licensed to Vium, Jupiter Orphan Therapeutics, Cohbar, Galilei Biosciences, GlaxoSmithKline, OvaScience, EMD Millipore, Wellomics, Inside Tracker, Caudalie, Bayer Crop Science, Longwood Fund, Zymo Research, EdenRoc Sciences (and affiliates Arc-Bio, Dovetail Genomics, Claret Bioscience, Revere Biosensors, UpRNA and MetroBiotech, Liberty Biosecurity). Life Biosciences (and affiliates Selphagy, Senolytic Therapeutics, Spotlight Biosciences, Animal Biosciences, Iduna, Immetas, Prana, Continuum Biosciences, Jumpstart Fertility, and Lua. D.A.S. sits on the board of directors of both companies. DAS is an inventor on a patent application licensed to Elysium Health. His personal royalty share is directed to the Sinclair lab.
Figures





References
-
- BAI P & CANTO C 2012. The role of PARP-1 and PARP-2 enzymes in metabolic regulation and disease. Cell Metab, 16, 290–5. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 CA190467/CA/NCI NIH HHS/United States
- R21 DE027490/DE/NIDCR NIH HHS/United States
- P30 DK056338/DK/NIDDK NIH HHS/United States
- R01 DK100263/DK/NIDDK NIH HHS/United States
- R01 AG019719/AG/NIA NIH HHS/United States
- R01 AG043483/AG/NIA NIH HHS/United States
- R01 DK098656/DK/NIDDK NIH HHS/United States
- P30 CA016672/CA/NCI NIH HHS/United States
- P30 CA125123/CA/NCI NIH HHS/United States
- R01 AG047924/AG/NIA NIH HHS/United States
- R37 AG028730/AG/NIA NIH HHS/United States
- DP1 AG058605/AG/NIA NIH HHS/United States
- R01 DK115454/DK/NIDDK NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

