Nkx-2.5 Regulates MDR1 Expression via Its Upstream Promoter in Breast Cancer Cells
- PMID: 30940996
- PMCID: PMC6439202
- DOI: 10.3346/jkms.2019.34.e100
Nkx-2.5 Regulates MDR1 Expression via Its Upstream Promoter in Breast Cancer Cells
Abstract
Background: Increased expression of MDR1 gene is one of the major mechanisms responsible for multidrug resistance in cancer cells. Two alternative promoters, upstream and downstream, are responsible for transcription of MDR1 gene in the human. However, the molecular mechanism regarding the transactivation of MDR1 upstream promoter (USP) has not been determined.
Methods: Dual-luciferase reporter gene assays were used to assess the effect of Nkx-2.5 on MDR1 USP activity using reporter plasmids for human MDR1 USP and its mutants. MDR1 mRNA level was examined by quantitative real-time PCR. The direct binding of Nkx-2.5 to the USP of MDR1 was evaluated by promoter enzyme immunoassays and chromatin immunoprecipitation assays.
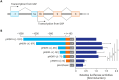
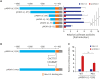
Results: Nkx-2.5 significantly stimulates the transactivation of MDR1 USP and increases MDR1 mRNA expression in MCF7 breast cancer cells. Reporter gene assays with deleted MDR1 USPs showed that the Nkx-2.5-binding site is located between positions -71 and +12. Mutation of the Nkx-2.5-binding site at nucleotide +4 to +10 markedly reduced the Nkx-2.5-mediated activation of MDR1 USP activity. A promoter binding immunoassay and a chromatin immunoprecipitation assay revealed that Nkx-2.5 binds directly to the region +4/+10 of human MDR1 USP.
Conclusion: The results in the present study show Nkx-2.5 is a positive regulator for the transactivation of MDR1 USP in MCF7 breast cancer cells. Our findings will help elucidate the regulatory mechanism responsible for the multidrug resistant cancer phenotype.
Keywords: Breast Cancer; MDR1; Nkx-2.5; Transcriptional Regulation.
Conflict of interest statement
Disclosure: The authors have no potential conflicts of interest to disclose.
Figures



References
-
- Juliano RL, Ling V. A surface glycoprotein modulating drug permeability in Chinese hamster ovary cell mutants. Biochim Biophys Acta. 1976;455(1):152–162. - PubMed
-
- Gottesman MM, Fojo T, Bates SE. Multidrug resistance in cancer: role of ATP-dependent transporters. Nat Rev Cancer. 2002;2(1):48–58. - PubMed
-
- Ikeda U, Ohkawa F, Seino Y, Yamamoto K, Hidaka Y, Kasahara T, et al. Serum interleukin 6 levels become elevated in acute myocardial infarction. J Mol Cell Cardiol. 1992;24(6):579–584. - PubMed
-
- Campos L, Guyotat D, Archimbaud E, Calmard-Oriol P, Tsuruo T, Troncy J, et al. Clinical significance of multidrug resistance P-glycoprotein expression on acute nonlymphoblastic leukemia cells at diagnosis. Blood. 1992;79(2):473–476. - PubMed
-
- Chan HS, Haddad G, Thorner PS, DeBoer G, Lin YP, Ondrusek N, et al. P-glycoprotein expression as a predictor of the outcome of therapy for neuroblastoma. N Engl J Med. 1991;325(23):1608–1614. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

