EXPRESS: Right Heart in Pulmonary Hypertension: From Adaptation to Failure
- PMID: 30942134
- PMCID: PMC6681271
- DOI: 10.1177/2045894019845611
EXPRESS: Right Heart in Pulmonary Hypertension: From Adaptation to Failure
Abstract
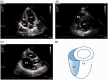
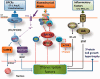
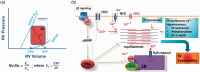
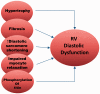
Right ventricular (RV) failure (RVF) has garnered significant attention in recent years because of its negative impact on clinical outcomes in patients with pulmonary hypertension (PH). PH triggers a series of events, including activation of several signaling pathways that regulate cell growth, metabolism, extracellular matrix remodeling, and energy production. These processes render the RV adaptive to PH. However, RVF develops when PH persists, accompanied by RV ischemia, alterations in substrate and mitochondrial energy metabolism, increased free oxygen radicals, increased cell loss, downregulation of adrenergic receptors, increased inflammation and fibrosis, and pathologic microRNAs. Diastolic dysfunction is also an integral part of RVF. Emerging non-invasive technologies such as molecular or metallic imaging, cardiac MRI, and ultrafast Doppler coronary flow mapping will be valuable tools to monitor RVF, especially the transition to RVF. Most PH therapies cannot treat RVF once it has occurred. A variety of therapies are available to treat acute and chronic RVF, but they are mainly supportive, and no effective therapy directly targets the failing RV. Therapies that target cell growth, cellular metabolism, oxidative stress, and myocyte regeneration are being tested preclinically. Future research should include establishing novel RVF models based on existing models, increasing use of human samples, creating human stem cell-based in vitro models, and characterizing alterations in cardiac excitation–contraction coupling during transition from adaptive RV to RVF. More successful strategies to manage RVF will likely be developed as we learn more about the transition from adaptive remodeling to maladaptive RVF in the future.
Figures

References
-
- Vonk-Noordegraaf A, Haddad F, Chin KM, et al. Right heart adaptation to pulmonary arterial hypertension: physiology and pathobiology. J Am Coll Cardiol 2013; 62: D22–33. - PubMed
-
- Haddad F, Doyle R, Murphy DJ, et al. Right ventricular function in cardiovascular disease, part II: pathophysiology, clinical importance, and management of right ventricular failure. Circulation 2008; 117: 1717–1731. - PubMed
-
- Galie N, Humbert M, Vachiery JL, et al. 2015 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension: The Joint Task Force for the Diagnosis and Treatment of Pulmonary Hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS): Endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC), International Society for Heart and Lung Transplantation (ISHLT). Eur Heart J 2016; 37: 67–119. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

