Neutrophil GM-CSF receptor dynamics in acute lung injury
- PMID: 30942918
- PMCID: PMC6850700
- DOI: 10.1002/JLB.3MA0918-347R
Neutrophil GM-CSF receptor dynamics in acute lung injury
Abstract
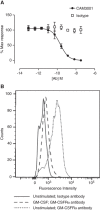
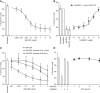
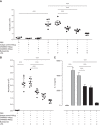
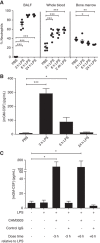
GM-CSF is important in regulating acute, persistent neutrophilic inflammation in certain settings, including lung injury. Ligand binding induces rapid internalization of the GM-CSF receptor (GM-CSFRα) complex, a process essential for signaling. Whereas GM-CSF controls many aspects of neutrophil biology, regulation of GM-CSFRα expression is poorly understood, particularly the role of GM-CSFRα in ligand clearance and whether signaling is sustained despite major down-regulation of GM-CSFRα surface expression. We established a quantitative assay of GM-CSFRα surface expression and used this, together with selective anti-GM-CSFR antibodies, to define GM-CSFRα kinetics in human neutrophils, and in murine blood and alveolar neutrophils in a lung injury model. Despite rapid sustained ligand-induced GM-CSFRα loss from the neutrophil surface, which persisted even following ligand removal, pro-survival effects of GM-CSF required ongoing ligand-receptor interaction. Neutrophils recruited to the lungs following LPS challenge showed initially high mGM-CSFRα expression, which along with mGM-CSFRβ declined over 24 hr; this was associated with a transient increase in bronchoalveolar lavage fluid (BALF) mGM-CSF concentration. Treating mice in an LPS challenge model with CAM-3003, an anti-mGM-CSFRα mAb, inhibited inflammatory cell influx into the lung and maintained the level of BALF mGM-CSF. Consistent with neutrophil consumption of GM-CSF, human neutrophils depleted exogenous GM-CSF, independent of protease activity. These data show that loss of membrane GM-CSFRα following GM-CSF exposure does not preclude sustained GM-CSF/GM-CSFRα signaling and that this receptor plays a key role in ligand clearance. Hence neutrophilic activation via GM-CSFR may play an important role in neutrophilic lung inflammation even in the absence of high GM-CSF levels or GM-CSFRα expression.
Keywords: LPS; alveolar; apoptosis; inflammation; signaling.
©2018 The Authors. Society for Leukocyte Biology Published by Wiley Periodicals, Inc.
Conflict of interest statement
All authors with MedImmune affiliation are (or were) employees of MedImmune and may have received AstraZeneca shares as part of their remuneration. There are no other conflicts of interest to declare.
Figures



References
-
- Thille AW, Esteban A, Fernandez‐Segoviano P. Chronology of histological lesions in acute respiratory distress syndrome with diffuse alveolar damage: a prospective cohort study of clinical autopsies. Lancet Respir Med. 2013;1:395‐401. - PubMed
-
- Weiland JE, Davis WB, Holter JF, Mohammed JR, Dorinsky PM, Gadek KE. Lung neutrophils in the adult respiratory distress syndrome. Clinical and pathophysiologic significance. Am Rev Respir Dis. 1986;133:218‐225. - PubMed
-
- Matute‐Bello G, Liles WC, Radella F. Neutrophil apoptosis in the acute respiratory distress syndrome. Am J Respir Crit Care Med. 1997;156:1969‐1977. - PubMed
-
- Root RK, Dale DC. Granulocyte colony‐stimulating factor and granulocyte‐macrophage colony‐stimulating factor: comparisons and potential for use in the treatment of infections in nonneutropenic patients. J Infect Dis. 1999;179:S342‐S352. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

