Dual role of the colonization factor CD2831 in Clostridium difficile pathogenesis
- PMID: 30944377
- PMCID: PMC6447587
- DOI: 10.1038/s41598-019-42000-8
Dual role of the colonization factor CD2831 in Clostridium difficile pathogenesis
Abstract
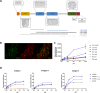
Clostridium difficile is a Gram-positive, anaerobic bacterium and the leading cause of antibiotic-associated diarrhea and pseudomembranous colitis. C. difficile modulates its transition from a motile to a sessile lifestyle through a mechanism of riboswitches regulated by cyclic diguanosine monophosphate (c-di-GMP). Previously described as a sortase substrate positively regulated by c-di-GMP, CD2831 was predicted to be a collagen-binding protein and thus potentially involved in sessility. By overexpressing CD2831 in C. difficile and heterologously expressing it on the surface of Lactococcus lactis, here we further demonstrated that CD2831 is a collagen-binding protein, able to bind to immobilized collagen types I, III and V as well as native collagen produced by human fibroblasts. We also observed that the overexpression of CD2831 raises the ability to form biofilm on abiotic surface in both C. difficile and L. lactis. Notably, we showed that CD2831 binds to the collagen-like domain of the human complement component C1q, suggesting a role in preventing complement cascade activation via the classical pathway. This functional characterization places CD2831 in the Microbial Surface Components Recognizing Adhesive Matrix Molecule (MSCRAMMs) family, a class of virulence factors with a dual role in adhesion to collagen-rich tissues and in host immune evasion by binding to human complement components.
Conflict of interest statement
This work was sponsored by Novartis Vaccines and Diagnostics Srl; in March 2015 the Novartis non-influenza Vaccines business was acquired by the GSK group of companies. V.A. is a student from The University of Padua, involved in a PhD studentship program at GSK. G.G., F.B., I.F., M.S., and R.L. were employees of Novartis Vaccines at the time of the study and are now employees of the GSK group of companies.
Figures




References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

