Amyloid-β oligomers have a profound detergent-like effect on lipid membrane bilayers, imaged by atomic force and electron microscopy
- PMID: 30948512
- PMCID: PMC6514634
- DOI: 10.1074/jbc.AC118.007195
Amyloid-β oligomers have a profound detergent-like effect on lipid membrane bilayers, imaged by atomic force and electron microscopy
Abstract
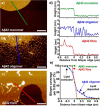
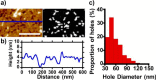
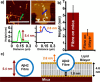
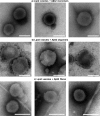
The ability of amyloid-β peptide (Aβ) to disrupt membrane integrity and cellular homeostasis is believed to be central to Alzheimer's disease pathology. Aβ is reported to have various impacts on the lipid bilayer, but a clearer picture of Aβ influence on membranes is required. Here, we use atomic force and transmission electron microscopies to image the impact of different isolated Aβ assembly types on lipid bilayers. We show that only oligomeric Aβ can profoundly disrupt the bilayer, visualized as widespread lipid extraction and subsequent deposition, which can be likened to an effect expected from the action of a detergent. We further show that Aβ oligomers cause widespread curvature and discontinuities within lipid vesicle membranes. In contrast, this detergent-like effect was not observed for Aβ monomers and fibers, although Aβ fibers did laterally associate and embed into the upper leaflet of the bilayer. The marked impact of Aβ oligomers on membrane integrity identified here reveals a mechanism by which these oligomers may be cytotoxic.
Keywords: Alzheimer disease; amyloid-beta (AB); atomic force microscopy (AFM); cytotoxicity; electron microscopy (EM); large unilamellar vesicles; membrane bilayer; membrane disruption; mica surface; nanoscale imaging; neurodegeneration; oligomer.
© 2019 Bode et al.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article
Figures

References
-
- Kuzyk A., Kastyak M., Agrawal V., Gallant M., Sivakumar G., Rak M., Del Bigio M. R., Westaway D., Julian R., and Gough K. M. (2010) Association among amyloid plaque, lipid, and creatine in hippocampus of TgCRND8 mouse model for Alzheimer disease. J. Biol. Chem. 285, 31202–31207 10.1074/jbc.M110.142174 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

