A Rat Model of Radiation Vasculitis for the Study of Mesenchymal Stem Cell-Based Therapy
- PMID: 30956979
- PMCID: PMC6431386
- DOI: 10.1155/2019/3727635
A Rat Model of Radiation Vasculitis for the Study of Mesenchymal Stem Cell-Based Therapy
Abstract
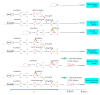
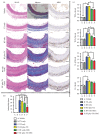
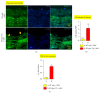
Radiation vasculitis is one of the most common detrimental effects of radiotherapy for malignant tumors. This is developed at the vasculature of adjacent organs. Animal experiments have showed that transplantation of mesenchymal stem cells (MSCs) restores vascular function after irradiation. But the population of MSCs being engrafted into irradiated vessels is too low in the conventional models to make assessment of therapeutic effect difficult. This is presumably because circulating MSCs are dispersed in adjacent tissues being irradiated simultaneously. Based on the assumption, a rat model, namely, RT (radiation) plus TX (transplantation), was established to promote MSC homing by sequestering irradiated vessels. In this model, a 1.5 cm long segment of rat abdominal aorta was irradiated by 160kV X-ray at a single dose of 35Gy before being procured and grafted to the healthy counterpart. F344 inbred rats served as both donors and recipients to exclude the possibility of immune rejection. A lead shield was used to confine X-ray delivery to a 3 cm×3 cm square-shaped field covering central abdominal region. The abdominal viscera especially small bowel and colon were protected from irradiation by being pushed off the central abdominal cavity. Typical radiation-induced vasculopathy was present on the 90th day after irradiation. The recruitment of intravenously injected MSCs to irradiated aorta was significantly improved by using the RT-plus-TX model as compared to the model with irradiation only. Generally, the RT-plus-Tx model promotes MSC recruitment to irradiated aorta by separating irradiated vascular segment from adjacent tissue. Thus, the model is preferred in the study of MSC-based therapy for radiation vasculitis when the evaluation of MSC homing is demanding.
Figures


References
-
- Harb A. H., Abou Fadel C., Sharara A. I. Radiation enteritis. Current Fungal Infection Reports. 2014;16(5):p. 383. - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

