Ponatinib Inhibits Proliferation and Induces Apoptosis of Liver Cancer Cells, but Its Efficacy Is Compromised by Its Activation on PDK1/Akt/mTOR Signaling
- PMID: 30959969
- PMCID: PMC6480565
- DOI: 10.3390/molecules24071363
Ponatinib Inhibits Proliferation and Induces Apoptosis of Liver Cancer Cells, but Its Efficacy Is Compromised by Its Activation on PDK1/Akt/mTOR Signaling
Abstract
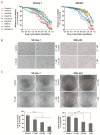
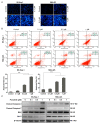
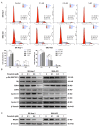
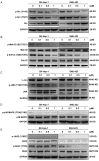
Ponatinib is a multi-target protein tyrosine kinase inhibitor, and its effects on hepatocellular carcinoma cells have not been previously explored. In the present study, we investigated its effects on hepatocellular carcinoma cell growth and the underlying mechanisms. Toward SK-Hep-1 and SNU-423 cells, ponatinib induces apoptosis by upregulation of cleaved caspase-3 and -7 and promotes cell cycle arrest in the G1 phase by inhibiting CDK4/6/CyclinD1 complex and phosphorylation of retinoblastoma protein. It inhibits the growth-stimulating mitogen-activated protein (MAP) kinase pathway, the phosphorylation of Src on both negative and positive regulation sites, and Jak2 and Stat3 phosphorylation. Surprisingly, it also activates the PDK1, the protein kinase B (Akt), and the mechanistic target of rapamycin (mTOR) signaling pathway. Blocking mTOR signaling strongly sensitizes cells to inhibition by ponatinib and makes ponatinib a much more potent inhibitor of hepatocellular carcinoma cell proliferation. These findings demonstrate that ponatinib exerts both positive and negative effects on hepatocellular cell proliferation, and eliminating its growth-stimulating effects by drug combination or potentially by chemical medication can significantly improve its efficacy as an anti-cancer drug.
Keywords: PDK1/Akt/mTOR signaling; apoptosis; liver cancer cells; ponatinib; proliferation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
[Ponatinib inhibits the proliferation of SNU-449 human hepatocellular cancer cells and blocks MAPK and PDK1/AKT/mTOR signaling pathways].Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2022 May;38(5):425-431. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2022. PMID: 35603651 Chinese.
-
Dasatinib inhibits proliferation of liver cancer cells, but activation of Akt/mTOR compromises dasatinib as a cancer drug.Acta Biochim Biophys Sin (Shanghai). 2021 Jul 5;53(7):823-836. doi: 10.1093/abbs/gmab061. Acta Biochim Biophys Sin (Shanghai). 2021. PMID: 33961012
-
PI3K/mTOR dual-inhibition with VS-5584 enhances anti-leukemic efficacy of ponatinib in blasts and Ph-negative LSCs of chronic myeloid leukemia.Eur J Pharmacol. 2021 Nov 5;910:174446. doi: 10.1016/j.ejphar.2021.174446. Epub 2021 Aug 27. Eur J Pharmacol. 2021. PMID: 34461124
-
Danusertib, a potent pan-Aurora kinase and ABL kinase inhibitor, induces cell cycle arrest and programmed cell death and inhibits epithelial to mesenchymal transition involving the PI3K/Akt/mTOR-mediated signaling pathway in human gastric cancer AGS and NCI-N78 cells.Drug Des Devel Ther. 2015 Mar 2;9:1293-318. doi: 10.2147/DDDT.S74964. eCollection 2015. Drug Des Devel Ther. 2015. PMID: 25767376 Free PMC article.
-
Ponatinib.Aust Prescr. 2015 Dec;38(6):221-2. doi: 10.18773/austprescr.2015.079. Epub 2015 Sep 18. Aust Prescr. 2015. PMID: 26843719 Free PMC article. Review. No abstract available.
Cited by
-
Discovery of Genomic Targets and Therapeutic Candidates for Liver Cancer Using Single-Cell RNA Sequencing and Molecular Docking.Biology (Basel). 2025 Apr 17;14(4):431. doi: 10.3390/biology14040431. Biology (Basel). 2025. PMID: 40282296 Free PMC article.
-
DepLink: an R Shiny app to systematically link genetic and pharmacologic dependencies of cancer.Bioinform Adv. 2023 Jun 12;3(1):vbad076. doi: 10.1093/bioadv/vbad076. eCollection 2023. Bioinform Adv. 2023. PMID: 37359725 Free PMC article.
-
Electrochemical and theoretical studies of the interaction between anticancer drug ponatinib and dsDNA.Sci Rep. 2024 Jan 27;14(1):2278. doi: 10.1038/s41598-024-52609-z. Sci Rep. 2024. PMID: 38280929 Free PMC article.
-
Integrated analysis and validation of the TRIM28-H2AX-CDK4 diagnostic model assists to predict the progression of HCC.Aging (Albany NY). 2023 Oct 20;15(24):14617-14650. doi: 10.18632/aging.205137. Epub 2023 Oct 20. Aging (Albany NY). 2023. PMID: 37870748 Free PMC article.
-
Gossypol enhances ponatinib's cytotoxicity against human hepatocellular carcinoma cells by involving cell cycle arrest, p-AKT/LC3II/p62, and Bcl2/caspase-3 pathways.Toxicol Rep. 2024 Dec 11;14:101856. doi: 10.1016/j.toxrep.2024.101856. eCollection 2025 Jun. Toxicol Rep. 2024. PMID: 39802605 Free PMC article.
References
-
- Fitzmaurice C., Allen C., Barber R.M., Barregard L., Bhutta Z.A., Brenner H., Dicker D.J., Chimed-Orchir O., Dandona R., Dandona L., et al. Global, Regional, and National Cancer Incidence, Mortality, Years of Life Lost, Years Lived with Disability, and Disability-Adjusted Life-years for 32 Cancer Groups, 1990 to 2015. JAMA Oncol. 2017;3:524. - PMC - PubMed
-
- Kimura T., Kato Y., Ozawa Y., Kodama K., Ito J., Ichikawa K., Yamada K., Hori Y., Tabata K., Takase K., et al. Immunomodulatory activity of lenvatinib contributes to antitumor activity in the Hepa1-6 hepatocellular carcinoma model. Cancer Sci. 2018;109:3993–4002. doi: 10.1111/cas.13806. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

