Intermolecular Interactions in Polyelectrolyte and Surfactant Complexes in Solution
- PMID: 30960035
- PMCID: PMC6401804
- DOI: 10.3390/polym11010051
Intermolecular Interactions in Polyelectrolyte and Surfactant Complexes in Solution
Abstract
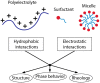
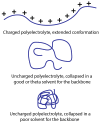
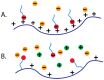
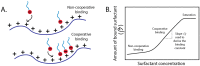
Polyelectrolytes are an important class of polymeric materials and are increasingly used in complex industrial formulations. A core use of these materials is in mixtures with surfactants, where a combination of hydrophobic and electrostatic interactions drives unique solution behavior and structure formation. In this review, we apply a molecular level perspective to the broad literature on polyelectrolyte-surfactant complexes, discussing explicitly the hydrophobic and electrostatic interaction contributions to polyelectrolyte surfactant complexes (PESCs), as well as the interplay between the two molecular interaction types. These interactions are sensitive to a variety of solution conditions, such as pH, ionic strength, mixing procedure, charge density, etc. and these parameters can readily be used to control the concentration at which structures form as well as the type of structure in the bulk solution.
Keywords: complexes; electrostatic; hydrophobic; micelle; molecular; polyelectrolyte; surfactant.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

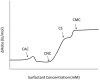
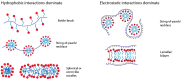
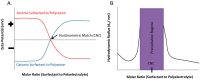
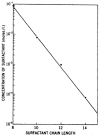


References
-
- Tadros T.F. An Introduction to Surfactants. Walter de Gruyter; Wokingham, UK: 2014.
-
- Rosen M., Kunjappu J. Surfactants and Interfacial Phenomena Index. 4th ed. John Wiley & Sons; Hoboken, NJ, USA: 2012.
-
- Rhein L.D., Schlossman M., O’Lenick A., Somasundaran P. Surfactants in Personal Care Products and Decorative Cosmetics. 3rd ed. CRC Press; Boca Raton, FL, USA: 2006.
-
- Van der Meeren P., Verstraetet W. Surfactants in relation to bioremediation and wastewater treatment. Curr. Opin. Colloid Interface Sci. 1996;1:624–634. doi: 10.1016/S1359-0294(96)80100-2. - DOI
Publication types
LinkOut - more resources
Full Text Sources

