Human Engineered Cartilage and Decellularized Matrix as an Alternative to Animal Osteoarthritis Model
- PMID: 30960663
- PMCID: PMC6403588
- DOI: 10.3390/polym10070738
Human Engineered Cartilage and Decellularized Matrix as an Alternative to Animal Osteoarthritis Model
Abstract
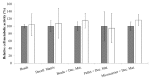
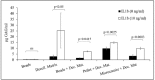
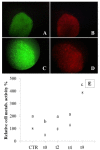
(1) Objective: to obtain a reproducible, robust, well-defined, and cost-affordable in vitro model of human cartilage degeneration, suitable for drug screening; (2) Methods: we proposed 3D models of engineered cartilage, considering two human chondrocyte sources (articular/nasal) and five culture methods (pellet, alginate beads, silk/alginate microcarriers, and decellularized cartilage). Engineered cartilages were treated with pro-inflammatory cytokine IL-1β to promote cartilage degradation; (3) Results: articular chondrocytes have been rejected since they exhibit low cellular doubling with respect to nasal cells, with longer culture time for cell expansion; furthermore, pellet and alginate bead cultures lead to insufficient cartilage matrix production. Decellularized cartilage resulted as good support for degeneration model, but long culture time and high cell amount are required to obtain the adequate scaffold colonization. Here, we proposed, for the first time, the combined use of decellularized cartilage, as aggrecanase substrate, with pellet, alginate beads, or silk/alginate microcarriers, as polymeric scaffolds for chondrocyte cultures. This approach enables the development of suitable models of cartilaginous pathology. The results obtained after cryopreservation also demonstrated that beads and microcarriers are able to preserve chondrocyte functionality and metabolic activity; (4) Conclusions: alginate and silk/alginate-based scaffolds can be easily produced and cryopreserved to obtain a cost-affordable and ready-to-use polymer-based product for the subsequent screening of anti-inflammatory drugs for cartilage diseases.
Keywords: alginate; beads; decellularized cartilage matrix; human chondrocytes; microcarrier; osteoarthritis; pellet; silk fibroin.
Conflict of interest statement
S.P. and M.L.T. are members of the advisory board of the company PharmaExceed S.r.l.
Figures




References
-
- Russell W.M.S., Burch R.L. The Principles of Humane Experimental Technique. Methuen; Wheathampstead, UK: 1959.
LinkOut - more resources
Full Text Sources

