Paradoxical Skin Reactions to Biologics in Patients With Rheumatologic Disorders
- PMID: 30971924
- PMCID: PMC6443901
- DOI: 10.3389/fphar.2019.00282
Paradoxical Skin Reactions to Biologics in Patients With Rheumatologic Disorders
Abstract
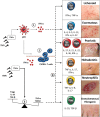
Targeted immune-modulating treatment with biological agents has revolutionized the management of immune-mediated inflammatory diseases, including rheumatologic conditions. The efficacy and tolerability of biological agents, from the initial tumour necrosis factor (TNF)-α inhibitors to the new anti-cytokine monoclonal antibodies, have dramatically changed the natural history of debilitating conditions such as rheumatoid arthritis and seronegative spondyloarthropathies. The widening use of biologics across several rheumatologic diseases has been associated with a new class of adverse events, the so-called paradoxical reactions. These events are inflammatory immune-mediated tissue reactions, developing paradoxically during treatment of rheumatologic conditions with targeted biologics that are commonly used for treating the idiopathic counterparts of these drug-induced reactions. The skin is frequently involved, and, even if considered rare to uncommon, these cutaneous manifestations are an important cause of biologic agent discontinuation. TNF-α antagonist-induced psoriasis, which can manifest de novo or as exacerbation of a pre-existing form, is the prototypic and most frequent paradoxical skin reaction to biologics while other reactions, such as eczematous and lichenoid eruptions, hidradenitis suppurativa, pyoderma gangrenosum, Sweet's syndrome and granulomatous skin diseases, occur much more rarely. Management of these reactions consists of topical or systemic skin-directed therapies, depending on the severity and extension of the cutaneous picture, and it is generally associated with switching over to other disease-modifying regimens for treating the underlying rheumatologic condition. Here, we review in detail the current concepts and controversies on classification, pathogenesis and clinical management of this new class of cutaneous adverse events induced by biologics in rheumatologic patients.
Keywords: TNFα-inhibitors; biologics; paradoxical skin reactions; psoriasis; rheumatological disorders.
Figures
References
-
- Brown G., Wang E., Leon A., Huynh M., Wehner M., Matro R., et al. (2017). Tumor necrosis factor-α inhibitor-induced psoriasis: systematic review of clinical features, histopathological findings, and management experience. J. Am. Acad. Dermatol. 76 334–341. 10.1016/j.jaad.2016.08.012 - DOI - PMC - PubMed

