The Osteogenic Potential of the Neural Crest Lineage May Contribute to Craniosynostosis
- PMID: 30976279
- PMCID: PMC6422151
- DOI: 10.1159/000493106
The Osteogenic Potential of the Neural Crest Lineage May Contribute to Craniosynostosis
Abstract
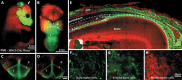
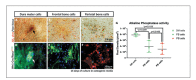
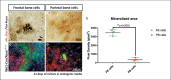
The craniofacial skeleton is formed from the neural crest and mesodermal lineages, both of which contribute mesenchymal precursors during formation of the skull bones. The large majority of cranial sutures also includes a proportion of neural crest-derived mesenchyme. While some studies have addressed the relative healing abilities of neural crest and mesodermal bone, relatively little attention has been paid to differences in intrinsic osteogenic potential. Here, we use mouse models to compare neural crest osteoblasts (from frontal bones or dura mater) to mesodermal osteoblasts (from parietal bones). Using in vitro culture approaches, we find that neural crest-derived osteoblasts readily generate bony nodules, while mesodermal osteoblasts do so less efficiently. Furthermore, we find that co-culture of neural crest-derived osteoblasts with mesodermal osteoblasts is sufficient to nucleate ossification centres. Altogether, this suggests that the intrinsic osteogenic abilities of neural crest-derived mesenchyme may be a primary driver behind craniosynostosis.
Keywords: Craniosynostosis; Dura mater; Neural crest; Osteogenesis; Sutures.
Figures


References
-
- Ang BU, Spivak RM, Nah HD, Kirschner RE. Dura in the pathogenesis of syndromic craniosynostosis: fibroblast growth factor receptor 2 mutations in dural cells promote osteogenic proliferation and differentiation of osteoblasts. J Craniofac Surg. 2010;21:462–467. - PubMed
-
- Bonilla F, Provencio M, Salas C, España P. Primary osteosarcoma of the meninges. Ann Oncol. 1994;5:965–966. - PubMed
-
- Boulet SL, Rasmussen SA, Honein MA. A population-based study of craniosynostosis in metropolitan Atlanta, 1989-2003. Am J Med Genet Part A. 2008;146A:984–991. - PubMed
-
- Bradley JP, Levine JP, Roth DA, McCarthy JG, Longaker MT. Studies in cranial suture biology: IV. Temporal sequence of posterior frontal cranial suture fusion in the mouse. Plast Reconstr Surg. 1996;98:1039–1045. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

