Oscillatory cortical forces promote three dimensional cell intercalations that shape the murine mandibular arch
- PMID: 30979871
- PMCID: PMC6461694
- DOI: 10.1038/s41467-019-09540-z
Oscillatory cortical forces promote three dimensional cell intercalations that shape the murine mandibular arch
Abstract
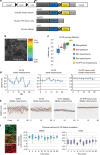
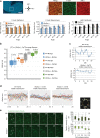
Multiple vertebrate embryonic structures such as organ primordia are composed of confluent cells. Although mechanisms that shape tissue sheets are increasingly understood, those which shape a volume of cells remain obscure. Here we show that 3D mesenchymal cell intercalations are essential to shape the mandibular arch of the mouse embryo. Using a genetically encoded vinculin tension sensor that we knock-in to the mouse genome, we show that cortical force oscillations promote these intercalations. Genetic loss- and gain-of-function approaches show that Wnt5a functions as a spatial cue to coordinate cell polarity and cytoskeletal oscillation. These processes diminish tissue rigidity and help cells to overcome the energy barrier to intercalation. YAP/TAZ and PIEZO1 serve as downstream effectors of Wnt5a-mediated actomyosin polarity and cytosolic calcium transients that orient and drive mesenchymal cell intercalations. These findings advance our understanding of how developmental pathways regulate biophysical properties and forces to shape a solid organ primordium.
Conflict of interest statement
The authors declare no competing interests.
Figures





References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

