Proinsulin C-Peptide Enhances Cell Survival and Protects against Simvastatin-Induced Myotoxicity in L6 Rat Myoblasts
- PMID: 30987105
- PMCID: PMC6479794
- DOI: 10.3390/ijms20071654
Proinsulin C-Peptide Enhances Cell Survival and Protects against Simvastatin-Induced Myotoxicity in L6 Rat Myoblasts
Abstract
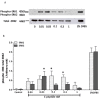
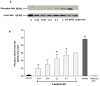
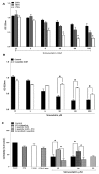
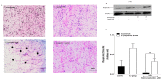
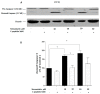
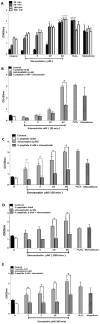
The repair capacity of progenitor skeletal muscle satellite cells (SC) in Type 1 diabetes mellitus (T1DM) is decreased. This is associated with the loss of skeletal muscle function. In T1DM, the deficiency of C-peptide along with insulin is associated with an impairment of skeletal muscle functions such as growth, and repair, and is thought to be an important contributor to increased morbidity and mortality. Recently, cholesterol-lowering drugs (statins) have also been reported to increase the risk of skeletal muscle dysfunction. We hypothesised that C-peptide activates key signaling pathways in myoblasts, thus promoting cell survival and protecting against simvastatin-induced myotoxicity. This was tested by investigating the effects of C-peptide on the L6 rat myoblast cell line under serum-starved conditions. Results: C-peptide at concentrations as low as 0.03 nM exerted stimulatory effects on intracellular signaling pathways-MAP kinase (ERK1/2) and Akt. When apoptosis was induced by simvastatin, 3 nM C-peptide potently suppressed the apoptotic effect through a pertussis toxin-sensitive pathway. Simvastatin strongly impaired Akt signaling and stimulated the reactive oxygen species (ROS) production; suggesting that Akt signaling and oxidative stress are important factors in statin-induced apoptosis in L6 myoblasts. The findings indicate that C-peptide exerts an important protective effect against death signaling in myoblasts. Therefore, in T1DM, the deficiency of C-peptide may contribute to myopathy by rendering myoblast-like progenitor cells (involved in muscle regeneration) more susceptible to the toxic effects of insults such as simvastatin.
Keywords: Type 1 diabetes; myotoxicity; proinsulin C-peptide; simvastatin.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Lysophosphatidic Acid Inhibits Simvastatin-Induced Myocytoxicity by Activating LPA Receptor/PKC Pathway.Molecules. 2020 Mar 27;25(7):1529. doi: 10.3390/molecules25071529. Molecules. 2020. PMID: 32230890 Free PMC article.
-
Simvastatin impairs ADP-stimulated respiration and increases mitochondrial oxidative stress in primary human skeletal myotubes.Free Radic Biol Med. 2012 Jan 1;52(1):198-207. doi: 10.1016/j.freeradbiomed.2011.10.449. Epub 2011 Oct 25. Free Radic Biol Med. 2012. PMID: 22080086 Free PMC article.
-
The AKT/mTOR signaling pathway plays a key role in statin-induced myotoxicity.Biochim Biophys Acta. 2015 Aug;1853(8):1841-9. doi: 10.1016/j.bbamcr.2015.04.010. Epub 2015 Apr 23. Biochim Biophys Acta. 2015. PMID: 25913013
-
The protective effect of Herba Cistanches on statin-induced myotoxicity in vitro.J Ethnopharmacol. 2016 Aug 22;190:68-73. doi: 10.1016/j.jep.2016.06.020. Epub 2016 Jun 7. J Ethnopharmacol. 2016. PMID: 27286913
-
C2C12 myoblasts are more sensitive to the toxic effects of simvastatin than myotubes and show impaired proliferation and myotube formation.Biochem Pharmacol. 2021 Aug;190:114649. doi: 10.1016/j.bcp.2021.114649. Epub 2021 Jun 7. Biochem Pharmacol. 2021. PMID: 34111424
Cited by
-
Diabetic Muscular Atrophy: Molecular Mechanisms and Promising Therapies.Front Endocrinol (Lausanne). 2022 Jun 30;13:917113. doi: 10.3389/fendo.2022.917113. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 35846289 Free PMC article. Review.
-
Utilize multi-metabolic parameters as determinants for prediction of skeletal muscle mass quality in elderly type2 diabetic Chinese patients.BMC Geriatr. 2024 Apr 9;24(1):325. doi: 10.1186/s12877-024-04827-3. BMC Geriatr. 2024. PMID: 38594634 Free PMC article.
-
β-Cell function is associated with osteosarcopenia in middle-aged and older nonobese patients with type 2 diabetes: A cross-sectional study.Open Med (Wars). 2021 Oct 20;16(1):1583-1590. doi: 10.1515/med-2021-0376. eCollection 2021. Open Med (Wars). 2021. PMID: 34722893 Free PMC article.
-
Does β-Hydroxy-β-Methylbutyrate Have Any Potential to Support the Treatment of Duchenne Muscular Dystrophy in Humans and Animals?Biomedicines. 2023 Aug 21;11(8):2329. doi: 10.3390/biomedicines11082329. Biomedicines. 2023. PMID: 37626825 Free PMC article. Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

