Tau repeat regions contain conserved histidine residues that modulate microtubule-binding in response to changes in pH
- PMID: 30992364
- PMCID: PMC6552421
- DOI: 10.1074/jbc.RA118.007004
Tau repeat regions contain conserved histidine residues that modulate microtubule-binding in response to changes in pH
Abstract
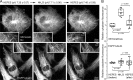
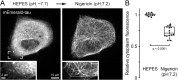
Tau, a member of the MAP2/tau family of microtubule-associated proteins, stabilizes and organizes axonal microtubules in healthy neurons. In neurodegenerative tauopathies, tau dissociates from microtubules and forms neurotoxic extracellular aggregates. MAP2/tau family proteins are characterized by three to five conserved, intrinsically disordered repeat regions that mediate electrostatic interactions with the microtubule surface. Here, we used molecular dynamics, microtubule-binding experiments, and live-cell microscopy, revealing that highly-conserved histidine residues near the C terminus of each microtubule-binding repeat are pH sensors that can modulate tau-microtubule interaction strength within the physiological intracellular pH range. We observed that at low pH (<7.5), these histidines are positively charged and interact with phenylalanine residues in a hydrophobic cleft between adjacent tubulin dimers. At higher pH (>7.5), tau deprotonation decreased binding to microtubules both in vitro and in cells. Electrostatic and hydrophobic characteristics of histidine were both required for tau-microtubule binding, as substitutions with constitutively and positively charged nonaromatic lysine or uncharged alanine greatly reduced or abolished tau-microtubule binding. Consistent with these findings, tau-microtubule binding was reduced in a cancer cell model with increased intracellular pH but was rapidly restored by decreasing the pH to normal levels. These results add detailed insights into the intracellular regulation of tau activity that may be relevant in both normal and pathological conditions.
Keywords: Tau protein (Tau); cancer biology; histidine; intracellular pH; intrinsically disordered protein; microtubule; microtubule-associated protein (MAP); molecular dynamics; neurobiology; neurodegeneration; neuronal cytoskeleton; pH sensing; protein–protein interaction.
© 2019 Charafeddine et al.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article
Figures





References
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

