Augmenter of liver regeneration protects the kidney from ischaemia-reperfusion injury in ferroptosis
- PMID: 30993878
- PMCID: PMC6533476
- DOI: 10.1111/jcmm.14302
Augmenter of liver regeneration protects the kidney from ischaemia-reperfusion injury in ferroptosis
Abstract
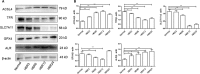
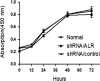
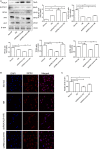
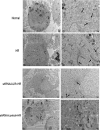
Acute kidney injury (AKI) is a common and severe clinical condition with high morbidity and mortality. Ischaemia-reperfusion (I/R) injury remains the major cause of AKI in the clinic. Ferroptosis is a recently discovered form of programmed cell death (PCD) that is characterized by iron-dependent accumulation of reactive oxygen species (ROS). Compelling evidence has shown that renal tubular cell death involves ferroptosis, although the underlying mechanisms remain unclear. Augmenter of liver regeneration (ALR) is a widely distributed multifunctional protein that is expressed in many tissues. Our previous study demonstrated that ALR possesses an anti-oxidant function. However, the modulatory mechanism of ALR remains unclear and warrants further investigation. Here, to elucidate the role of ALR in ferroptosis, ALR expression was inhibited using short hairpin RNA lentivirals (shRNA) in vitro model of I/R-induced AKI. The results suggest that the level of ferroptosis is increased, particularly in the shRNA/ALR group, accompanied by increased ROS and mitochondrial damage. Furthermore, inhibition of system xc- with erastin aggravates ferroptosis, particularly silencing of the expression of ALR. Unexpectedly, we demonstrate a novel signalling pathway of ferroptosis. In summary, we show for the first time that silencing ALR aggravates ferroptosis in an in vitro model of I/R. Notably, we show that I/R induced kidney ferroptosis is mediated by ALR, which is linked to the glutathione-glutathione peroxidase (GSH-GPx) system.
Keywords: acute kidney injury (AKI); augmenter of liver regeneration (ALR); ferroptosis; ischaemia-reperfusion (I/R) injury; reactive oxygen species (ROS).
© 2019 The Authors. Journal of Cellular and Molecular Medicine published by John Wiley & Sons Ltd and Foundation for Cellular and Molecular Medicine.
Conflict of interest statement
The authors have no conflicts of interest or financial interests to declare.
Figures



References
-
- Hoste EA, Bagshaw SM, Bellomo R, et al. Epidemiology of acute kidney injury in critically ill patients: the multinational AKI‐EPI study. Intensive Care Med. 2015;41:1411‐1423. - PubMed
-
- Mehta RL, Burdmann EA, Cerdá J, et al. Recognition and management of acute kidney injury in the International Society of Nephrology 0by25 Global Snapshot: a multinational cross‐sectional study. The Lancet. 2016;387:2017‐2025. - PubMed
-
- Liaño F, Pascual J, The Madrid Acute Renal Failure Study G . Epidemiology of acute renal failure: a prospective, multicenter, community‐based study. Kidney Int. 1996;50:811‐818. - PubMed
-
- Kim J, Jung KJ, Park KM. Reactive oxygen species differently regulate renal tubular epithelial and interstitial cell proliferation after ischemia and reperfusion injury. Am J Physiol Renal Physiol. 2010;298:F1118‐F1129. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

