Phospho-valproic acid (MDC-1112) suppresses glioblastoma growth in preclinical models through the inhibition of STAT3 phosphorylation
- PMID: 30994173
- PMCID: PMC7967702
- DOI: 10.1093/carcin/bgz069
Phospho-valproic acid (MDC-1112) suppresses glioblastoma growth in preclinical models through the inhibition of STAT3 phosphorylation
Abstract
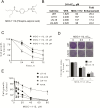
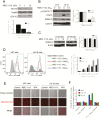
New therapeutic strategies against glioblastoma multiforme (GBM) are urgently needed. Signal transducer and activator of transcription 3 (STAT3), constitutively active in many GBM tumors, plays a major role in GBM tumor growth and represents a potential therapeutic target. We have documented previously that phospho-valproic acid (MDC-1112), which inhibits STAT3 activation, possesses strong anticancer properties in multiple cancer types. In this study, we explored the anticancer efficacy of MDC-1112 in preclinical models of GBM, and evaluated its mode of action. MDC-1112 inhibited the growth of multiple human GBM cell lines in a concentration- and time-dependent manner. Normal human astrocytes were resistant to MDC-1112, indicating selectivity. In vivo, MDC-1112 reduced the growth of subcutaneous GBM xenografts in mice by up to 78.2% (P < 0.01), compared with the controls. Moreover, MDC-1112 extended survival in an intracranial xenograft model. Although all vehicle-treated mice died by 19 days of treatment, 7 of 11 MDC-1112-treated mice were alive and healthy by the end of 5 weeks, with many showing tumor regression. Mechanistically, MDC-1112 inhibited STAT3 phosphorylation at the serine 727 residue, but not at tyrosine 705, in vitro and in vivo. STAT3 overexpression rescued GBM cells from the cell growth inhibition by MDC-1112. In addition, MDC-1112 reduced STAT3 levels in the mitochondria and enhanced mitochondrial levels of reactive oxygen species, which triggered apoptosis. In conclusion, MDC-1112 displays strong efficacy in preclinical models of GBM, with the serine 727 residue of STAT3 being its key molecular target. MDC-1112 merits further evaluation as a drug candidate for GBM. New therapeutic options are needed for glioblastoma. The novel agent MDC-1112 is an effective anticancer agent in multiple animal models of glioblastoma, and its mechanism of action involves the inhibition of STAT3 phosphorylation, primarily at its Serine 727 residue.
© The Author(s) 2019. Published by Oxford University Press. All rights reserved. For Permissions, please email: journals.permissions@oup.com.
Figures




References
-
- Tykocki, T. et al. (2018) Ten-year survival in glioblastoma. A systematic review. J. Clin. Neurosci., 54, 7–13. - PubMed
-
- Nagasawa, D.T. et al. (2012) Temozolomide and other potential agents for the treatment of glioblastoma multiforme. Neurosurg. Clin. N. Am., 23, 307–22, ix. - PubMed
-
- Stupp, R. et al. ; European Organisation for Research and Treatment of Cancer Brain Tumor and Radiotherapy Groups; National Cancer Institute of Canada Clinical Trials Group. (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N. Engl. J. Med., 352, 987–996. - PubMed
-
- Kim, B.H. et al. (2016) Signal transducer and activator of transcription 3 as a therapeutic target for cancer and the tumor microenvironment. Arch. Pharm. Res., 39, 1085–1099. - PubMed

