Phase III Trial: Adjuvant Pelvic Radiation Therapy Versus Vaginal Brachytherapy Plus Paclitaxel/Carboplatin in High-Intermediate and High-Risk Early Stage Endometrial Cancer
- PMID: 30995174
- PMCID: PMC6804858
- DOI: 10.1200/JCO.18.01575
Phase III Trial: Adjuvant Pelvic Radiation Therapy Versus Vaginal Brachytherapy Plus Paclitaxel/Carboplatin in High-Intermediate and High-Risk Early Stage Endometrial Cancer
Abstract
Purpose: The primary objective was to determine if vaginal cuff brachytherapy and chemotherapy (VCB/C) increases recurrence-free survival (RFS) compared with pelvic radiation therapy (RT) in high-intermediate and high-risk early-stage endometrial carcinoma.
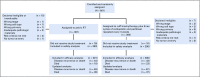
Patients and methods: A randomized phase III trial was performed in eligible patients with endometrial cancer. Eligible patients had International Federation of Gynecology and Obstetrics (2009) stage I endometrioid histology with Gynecologic Oncology Group protocol 33-based high-intermediate-risk criteria, stage II disease, or stage I to II serous or clear cell tumors. Treatment was randomly assigned between RT (45 to 50.4 Gy over 5 weeks) or VCB followed by intravenous paclitaxel 175 mg/m2 (3 hours) plus carboplatin (area under the curve, 6) every 21 days for three cycles.
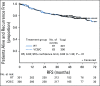
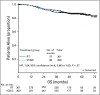
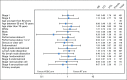
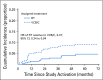
Results: The median age of the 601 patients was 63 years, and 74% had stage I disease. Histologies included endometrioid (71%), serous (15%), and clear cell (5%). With a median follow-up of 53 months, the 60-month RFS was 0.76 (95% CI, 0.70 to 0.81) for RT and 0.76 (95% CI, 0.70 to 0.81) for VCB/C (hazard ratio, 0.92; 90% confidence limit, 0.69 to 1.23). The 60-month overall survival was 0.87 (95% CI, 0.83 to 0.91) for RT and 0.85 (95% CI, 0.81 to 0.90) for VCB/C (hazard ratio, 1.04; 90% confidence limit, 0.71 to 1.52). Vaginal and distant recurrence rates were similar between arms. Pelvic or para-aortic nodal recurrences were more common with VCB/C (9% v 4%). There was no heterogeneity of treatment effect with respect to RFS or overall survival among clinical or pathologic variables evaluated.
Conclusion: Superiority of VCB/C compared with pelvic RT was not demonstrated. Acute toxicity was greater with VCB/C; late toxicity was similar. Pelvic RT alone remains an effective, well-tolerated, and appropriate adjuvant treatment in high-risk early-stage endometrial carcinomas of all histologies.
Trial registration: ClinicalTrials.gov NCT00807768.
Figures

Comment in
-
External Beam, Brachytherapy, or Chemotherapy? Defining Adjuvant Therapy for Early-Stage and High- and High-Intermediate-Risk Endometrial Cancer.J Clin Oncol. 2019 Jul 20;37(21):1778-1784. doi: 10.1200/JCO.19.00362. Epub 2019 Jun 4. J Clin Oncol. 2019. PMID: 31163010
References
-
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018. CA Cancer J Clin. 2018;68:7–30. - PubMed
-
- Aalders J, Abeler V, Kolstad P, et al. Postoperative external irradiation and prognostic parameters in stage I endometrial carcinoma: Clinical and histopathologic study of 540 patients. Obstet Gynecol. 1980;56:419–427. - PubMed
-
- Creutzberg CL, van Putten WL, Koper PC, et al. Surgery and postoperative radiotherapy versus surgery alone for patients with stage-1 endometrial carcinoma: Multicentre randomised trial—PORTEC Study Group: Post operative radiation therapy in endometrial carcinoma. Lancet. 2000;355:1404–1411. - PubMed
-
- Keys HM, Roberts JA, Brunetto VL, et al. A phase III trial of surgery with or without adjunctive external pelvic radiation therapy in intermediate risk endometrial adenocarcinoma: A Gynecologic Oncology Group study. Gynecol Oncol. 2004;92:744–751. - PubMed
-
- Kong A, Johnson N, Kitchener HC, et al. Adjuvant radiotherapy for stage I endometrial cancer: An updated Cochrane systematic review and meta-analysis. J Natl Cancer Inst. 2012;104:1625–1634. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

