Intracellular Peptides in Cell Biology and Pharmacology
- PMID: 30995799
- PMCID: PMC6523763
- DOI: 10.3390/biom9040150
Intracellular Peptides in Cell Biology and Pharmacology
Abstract
Intracellular peptides are produced by proteasomes following degradation of nuclear, cytosolic, and mitochondrial proteins, and can be further processed by additional peptidases generating a larger pool of peptides within cells. Thousands of intracellular peptides have been sequenced in plants, yeast, zebrafish, rodents, and in human cells and tissues. Relative levels of intracellular peptides undergo changes in human diseases and also when cells are stimulated, corroborating their biological function. However, only a few intracellular peptides have been pharmacologically characterized and their biological significance and mechanism of action remains elusive. Here, some historical and general aspects on intracellular peptides' biology and pharmacology are presented. Hemopressin and Pep19 are examples of intracellular peptides pharmacologically characterized as inverse agonists to cannabinoid type 1 G-protein coupled receptors (CB1R), and hemopressin fragment NFKF is shown herein to attenuate the symptoms of pilocarpine-induced epileptic seizures. Intracellular peptides EL28 (derived from proteasome 26S protease regulatory subunit 4; Rpt2), PepH (derived from Histone H2B type 1-H), and Pep5 (derived from G1/S-specific cyclin D2) are examples of peptides that function intracellularly. Intracellular peptides are suggested as biological functional molecules, and are also promising prototypes for new drug development.
Keywords: cancer; drug discovery; endocannabinoid; epilepsy; insulin resistance; intracellular peptides; obesity; proteasome.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


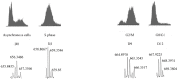
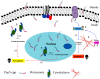
References
-
- Lee C., Zeng J., Drew B.G., Sallam T., Martin-Montalvo A., Wan J., Kim S.J., Mehta H., Hevener A.L., de Cabo R., et al. The mitochondrial-derived peptide MOTS-c promotes metabolic homeostasis and reduces obesity and insulin resistance. Cell Metab. 2015;21:443–454. doi: 10.1016/j.cmet.2015.02.009. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

