Myelin repair stimulated by CNS-selective thyroid hormone action
- PMID: 30996143
- PMCID: PMC6538346
- DOI: 10.1172/jci.insight.126329
Myelin repair stimulated by CNS-selective thyroid hormone action
Abstract
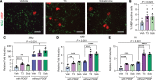
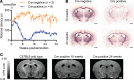
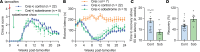
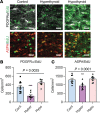
Oligodendrocyte processes wrap axons to form neuroprotective myelin sheaths, and damage to myelin in disorders, such as multiple sclerosis (MS), leads to neurodegeneration and disability. There are currently no approved treatments for MS that stimulate myelin repair. During development, thyroid hormone (TH) promotes myelination through enhancing oligodendrocyte differentiation; however, TH itself is unsuitable as a remyelination therapy due to adverse systemic effects. This problem is overcome with selective TH agonists, sobetirome and a CNS-selective prodrug of sobetirome called Sob-AM2. We show here that TH and sobetirome stimulated remyelination in standard gliotoxin models of demyelination. We then utilized a genetic mouse model of demyelination and remyelination, in which we employed motor function tests, histology, and MRI to demonstrate that chronic treatment with sobetirome or Sob-AM2 leads to significant improvement in both clinical signs and remyelination. In contrast, chronic treatment with TH in this model inhibited the endogenous myelin repair and exacerbated disease. These results support the clinical investigation of selective CNS-penetrating TH agonists, but not TH, for myelin repair.
Keywords: Endocrinology; Mouse models; Multiple sclerosis; Neuroscience.
Conflict of interest statement
Figures



References
-
- Barres BA, Lazar MA, Raff MC. A novel role for thyroid hormone, glucocorticoids and retinoic acid in timing oligodendrocyte development. Development. 1994;120(5):1097–1108. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

