Development and Application of a Functional Human Esophageal Mucosa Explant Platform to Eosinophilic Esophagitis
- PMID: 30996235
- PMCID: PMC6470157
- DOI: 10.1038/s41598-019-41147-8
Development and Application of a Functional Human Esophageal Mucosa Explant Platform to Eosinophilic Esophagitis
Abstract
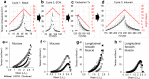
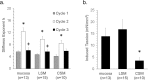
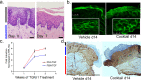
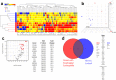
There is an increasing prevalence of esophageal diseases but intact human tissue platforms to study esophageal function, disease mechanisms, and the interactions between cell types in situ are lacking. To address this, we utilized full thickness human donor esophagi to create and validate the ex vivo function of mucosa and smooth muscle (n = 25). Explanted tissue was tested for contractile responses to carbachol and histamine. We then treated ex vivo human esophageal mucosa with a cytokine cocktail to closely mimic the Th2 and inflammatory milieu of eosinophilic esophagitis (EoE) and assessed alterations in smooth muscle and extracellular matrix function and stiffening. We found that full thickness human esophagus as well as the individual layers of circular and longitudinal muscularis propria developed tension in response to carbachol ex vivo and that mucosa demonstrated squamous cell differentiation. Treatment of mucosa with Th2 and fibrotic cytokines recapitulated the majority of the clinical Eosinophilic Esophagitis Diagnostic Profile (EDP) on fluidic transcriptional microarray. Transforming growth factor-beta-1 (TGFβ1) increased gene expression of fibronectin, smooth muscle actin, and phospholamban (p < 0.001). The EoE cocktail also increased stiffness and decreased mucosal compliance, akin to the functional alterations in EoE (p = 0.001). This work establishes a new, transcriptionally intact and physiologically functional human platform to model esophageal tissue responses in EoE.
Conflict of interest statement
The authors declare no competing interests.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
- AI135034/Division of Intramural Research, National Institute of Allergy and Infectious Diseases (Division of Intramural Research of the NIAID)/International
- UL1 TR000039/TR/NCATS NIH HHS/United States
- R01 DK114457/DK/NIDDK NIH HHS/United States
- R01 AI092135/AI/NIAID NIH HHS/United States
- K24 AI135034/AI/NIAID NIH HHS/United States
- U19 AI070235/AI/NIAID NIH HHS/United States
- R01 AI124355/AI/NIAID NIH HHS/United States
- AI091235/Division of Intramural Research, National Institute of Allergy and Infectious Diseases (Division of Intramural Research of the NIAID)/International
- R25 GM083247/GM/NIGMS NIH HHS/United States
- AI070235/U.S. Department of Health & Human Services | NIH | National Institute of Allergy and Infectious Diseases (NIAID)/International
- M01 RR000080/RR/NCRR NIH HHS/United States
- R37 AI045898/AI/NIAID NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

