Deciphering the intersystem crossing in near-infrared BODIPY photosensitizers for highly efficient photodynamic therapy
- PMID: 30996892
- PMCID: PMC6429462
- DOI: 10.1039/c8sc04840a
Deciphering the intersystem crossing in near-infrared BODIPY photosensitizers for highly efficient photodynamic therapy
Abstract
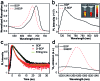
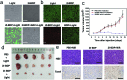
Deciphering singlet-to-triplet intersystem crossing (ISC) in organic near-infrared photosensitizers (PSs) is of fundamental importance in the designing of high-performance PSs to boost the clinical usage of photodynamic therapy (PDT). However, in-depth investigations of the ISC dynamics in near-infrared PSs have not been performed to date. Here, systematical investigations of the ISC dynamics in organic near-infrared BODIPY derivatives are presented, in which a multi-channel yet remarkably efficient ISC process is revealed by ultrafast femtosecond transient absorption (fs-TA) spectroscopy and theoretical calculation. The fs-TA verifies an exceptionally enhanced ISC efficiency (Φ ISC = 91%) in iodine-substituted BODIPY (2I-BDP) which is further supported by the calculation results. This endows 2I-BDP with an ultrahigh singlet oxygen quantum yield (Φ Δ = 88%), thus enabling a proof-of-concept application of highly efficient PDT in vivo under ultralow near-infrared light power density (10 mW cm-2). The in-depth understanding of ISC dynamics in organic near-infrared materials may provide valuable guidance in the designing of novel organic theranostic materials for clinical cancer treatment.
Figures


Similar articles
-
Manipulating the Dynamics of Dark Excited States in Organic Materials for Phototheranostics.Acc Chem Res. 2021 Feb 2;54(3):697-706. doi: 10.1021/acs.accounts.0c00688. Epub 2020 Dec 10. Acc Chem Res. 2021. PMID: 33301301
-
Spin-Orbit Charge-Transfer Intersystem Crossing (ISC) in Compact Electron Donor-Acceptor Dyads: ISC Mechanism and Application as Novel and Potent Photodynamic Therapy Reagents.Chemistry. 2020 Jan 22;26(5):1091-1102. doi: 10.1002/chem.201904306. Epub 2020 Jan 16. Chemistry. 2020. PMID: 31743947
-
Design and Synthesis of BODIPY-Hetero[5]helicenes as Heavy-Atom-Free Triplet Photosensitizers for Photodynamic Therapy of Cancer.Chemistry. 2023 Oct 13;29(57):e202301605. doi: 10.1002/chem.202301605. Epub 2023 Jul 28. Chemistry. 2023. PMID: 37314387
-
Bodipy Derivatives as Triplet Photosensitizers and the Related Intersystem Crossing Mechanisms.Front Chem. 2019 Dec 12;7:821. doi: 10.3389/fchem.2019.00821. eCollection 2019. Front Chem. 2019. PMID: 31921760 Free PMC article. Review.
-
Stimuli-Responsive Reversible Switching of Intersystem Crossing in Pure Organic Material for Smart Photodynamic Therapy.Angew Chem Int Ed Engl. 2019 Aug 5;58(32):11105-11111. doi: 10.1002/anie.201905129. Epub 2019 Jul 3. Angew Chem Int Ed Engl. 2019. PMID: 31172619 Review.
Cited by
-
Molecular Design of Conjugated Small Molecule Nanoparticles for Synergistically Enhanced PTT/PDT.Nanomicro Lett. 2020 Jul 13;12(1):147. doi: 10.1007/s40820-020-00474-6. Nanomicro Lett. 2020. PMID: 34138129 Free PMC article.
-
A lysosomes and mitochondria dual-targeting AIE-active NIR photosensitizer: Constructing amphiphilic structure for enhanced antitumor activity and two-photon imaging.Mater Today Bio. 2023 Jul 5;21:100721. doi: 10.1016/j.mtbio.2023.100721. eCollection 2023 Aug. Mater Today Bio. 2023. PMID: 37502829 Free PMC article.
-
Exploiting a Neutral BODIPY Copolymer as an Effective Agent for Photodynamic Antimicrobial Inactivation.J Phys Chem B. 2021 Feb 18;125(6):1550-1557. doi: 10.1021/acs.jpcb.0c09634. Epub 2021 Feb 4. J Phys Chem B. 2021. PMID: 33538173 Free PMC article.
-
Elucidating heterogeneous photocatalytic superiority of microporous porphyrin organic cage.Nat Commun. 2020 Feb 26;11(1):1047. doi: 10.1038/s41467-020-14831-x. Nat Commun. 2020. PMID: 32103004 Free PMC article.
-
High- and Low-Fluorescent Photoinitiators for Multiphoton Lithography.ACS Appl Polym Mater. 2025 Jul 31;7(15):10108-10120. doi: 10.1021/acsapm.5c01802. eCollection 2025 Aug 8. ACS Appl Polym Mater. 2025. PMID: 40809104 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

