Nanolayered cobalt-molybdenum sulphides (Co-Mo-S) catalyse borrowing hydrogen C-S bond formation reactions of thiols or H2S with alcohols
- PMID: 30996896
- PMCID: PMC6429612
- DOI: 10.1039/c8sc05782f
Nanolayered cobalt-molybdenum sulphides (Co-Mo-S) catalyse borrowing hydrogen C-S bond formation reactions of thiols or H2S with alcohols
Abstract
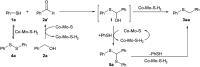
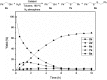
Nanolayered cobalt-molybdenum sulphide (Co-Mo-S) materials have been established as excellent catalysts for C-S bond construction. These catalysts allow for the preparation of a broad range of thioethers in good to excellent yields from structurally diverse thiols and readily available primary as well as secondary alcohols. Chemoselectivity in the presence of sensitive groups such as double bonds, nitriles, carboxylic esters and halogens has been demonstrated. It is also shown that the reaction takes place through a hydrogen-autotransfer (borrowing hydrogen) mechanism that involves Co-Mo-S-mediated dehydrogenation and hydrogenation reactions. A novel catalytic protocol based on the thioetherification of alcohols with hydrogen sulphide (H2S) to furnish symmetrical thioethers has also been developed using these earth-abundant metal-based sulphide catalysts.
Figures
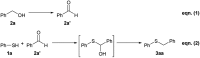

Similar articles
-
Alloying Engineering of Defective Molybdenum Sulfide Basal Planes for Enhanced Borrowing Hydrogen Activity in the Thioetherification of Alcohols.ChemSusChem. 2025 Jun 2;18(11):e202500343. doi: 10.1002/cssc.202500343. Epub 2025 Apr 3. ChemSusChem. 2025. PMID: 40080409 Free PMC article.
-
3d-Metal Catalyzed N- and C-Alkylation Reactions via Borrowing Hydrogen or Hydrogen Autotransfer.Chem Rev. 2019 Feb 27;119(4):2524-2549. doi: 10.1021/acs.chemrev.8b00306. Epub 2018 Nov 20. Chem Rev. 2019. PMID: 30457320
-
Cobalt-Catalyzed Alkylation of Secondary Alcohols with Primary Alcohols via Borrowing Hydrogen/Hydrogen Autotransfer.Chemistry. 2017 Sep 7;23(50):12110-12113. doi: 10.1002/chem.201701211. Epub 2017 May 19. Chemistry. 2017. PMID: 28452422
-
Manganese-catalyzed C-C and C-N bond formation with alcohols via borrowing hydrogen or hydrogen auto-transfer.Beilstein J Org Chem. 2024 May 21;20:1111-1166. doi: 10.3762/bjoc.20.98. eCollection 2024. Beilstein J Org Chem. 2024. PMID: 38887586 Free PMC article. Review.
-
C-Alkylation of Ketones and Related Compounds by Alcohols: Transition-Metal-Catalyzed Dehydrogenation.Angew Chem Int Ed Engl. 2016 Jan 18;55(3):862-75. doi: 10.1002/anie.201507521. Epub 2015 Dec 7. Angew Chem Int Ed Engl. 2016. PMID: 26639633 Review.
Cited by
-
Alloying Engineering of Defective Molybdenum Sulfide Basal Planes for Enhanced Borrowing Hydrogen Activity in the Thioetherification of Alcohols.ChemSusChem. 2025 Jun 2;18(11):e202500343. doi: 10.1002/cssc.202500343. Epub 2025 Apr 3. ChemSusChem. 2025. PMID: 40080409 Free PMC article.
-
Chemoselective Heterogeneous Hydrogenation of Sulfur Containing Quinolines under Mild Conditions.J Am Chem Soc. 2024 Mar 6;146(9):5864-5871. doi: 10.1021/jacs.3c11163. Epub 2024 Feb 20. J Am Chem Soc. 2024. PMID: 38378184 Free PMC article.
-
Functionalization of Electron-Rich Secondary Benzyl Alcohols in HFIP: From Thioethers to Trisubstituted Methanes.J Org Chem. 2025 Jul 11;90(27):9513-9518. doi: 10.1021/acs.joc.5c00900. Epub 2025 Jun 28. J Org Chem. 2025. PMID: 40580135 Free PMC article.
-
Access to thioethers from thiols and alcohols via homogeneous and heterogeneous catalysis.Sci Rep. 2023 Nov 23;13(1):20624. doi: 10.1038/s41598-023-47938-4. Sci Rep. 2023. PMID: 37996490 Free PMC article.
-
Molecularly Engineering Defective Basal Planes in Molybdenum Sulfide for the Direct Synthesis of Benzimidazoles by Reductive Coupling of Dinitroarenes with Aldehydes.JACS Au. 2022 Feb 9;2(3):601-612. doi: 10.1021/jacsau.1c00477. eCollection 2022 Mar 28. JACS Au. 2022. PMID: 35373204 Free PMC article.
References
-
- Block E., Reactions of Organosulfur Compounds, Academic Press, New York, 1978.
- Peach M. E., in The Chemistry of the Thiol Group, ed. S. Patai, John Wiley & Sons, London, 1979, pp. 721–723.
- Bernardi F., Csizmadia I. G. and Mangini A., Organic Sulfur Chemistry. Theoretical and Experimental Advances, Elsevier, Amsterdam, 1985.
- Cremlyn R. J., An Introduction to Organosulfur Chemistry, John Wiley & Sons, New York, 1996.
- Kondo T., Mitsudo T.-a. Chem. Rev. 2000;100:3205–3220. - PubMed
- Liu H., Jiang X. Chem.–Asian J. 2013;8:2546–2563. - PubMed
-
- Artico M., Silvestri R., Pagnozzi E., Bruno B., Novellino E., Greco G., Massa S., Ettorre A., Loi A. G., Scintu F., La Colla P. J. Med. Chem. 2000;43:1886–1891. - PubMed
- Sun Z.-Y., Botros E., Su A.-D., Kim Y., Wang E., Baturay N. Z., Kwon C.-H. J. Med. Chem. 2000;43:4160–4168. - PubMed
- Wang Y., Chackalamannil S., Chang W., Greenlee W., Ruperto V., Duffy R. A., McQuade R., Lachowicz J. E. Bioorg. Med. Chem. Lett. 2001;11:891–894. - PubMed
- Faucher A.-M., White P. W., Brochu C., Grand-Maître C., Rancourt J., Fazal G. J. Med. Chem. 2004;47:18–21. - PubMed
- Clader J. W., Billard W., Binch H., Chen L.-Y., Crosby G., Duffy R. A., Ford J., Kozlowski J. A., Lachowicz J. E., Li S., Liu C., McCombie S. W., Vice S., Zhou G., Greenlee W. J. Biorg. Med. Chem. 2004;12:319–326. - PubMed
- Sciabola S., Carosati E., Baroni M., Mannhold R. J. Med. Chem. 2005;48:3756–3767. - PubMed
- Gangjee A., Zeng Y., Talreja T., McGuire J. J., Kisliuk R. L., Queener S. F. J. Med. Chem. 2007;50:3046–3053. - PMC - PubMed
- Minghao F., Bingqing T., Steven H. L. a. X. J. Curr. Top. Med. Chem. 2016;16:1200–1216. - PubMed
-
- Glass H. B., Reid E. E. J. Am. Chem. Soc. 1929;51:3428–3430.
- Dougherty G., Hammond P. D. J. Am. Chem. Soc. 1935;57:117–118.
- Kharasch N., Potempa S. J., Wehrmeister H. L. Chem. Rev. 1946;39:269–332. - PubMed
- Parham W. E., Wynberg H. Org. Synth. 1955;35:51.
- Patai S., The Chemistry of the Functional Groups – The Chemistry of the Thiol Group, Wiley, London, 1974.
- Herriott A. W., Picker D. J. Am. Chem. Soc. 1975;97:2345–2349.
- Landini D., Rolla F. Org. Synth. 1978;58:143.
- Boscato J. F., Catala J. M., Franta E., Brossas J. Tetrahedron Lett. 1980;21:1519–1520.
- Kosugi M., Ogata T., Terada M., Sano H., Migita T. Bull. Chem. Soc. Jpn. 1985;58:3657–3658.
- Hundscheid F. J. A., Tandon V. K., Rouwette P. H. F. M., van Leusen A. M. Tetrahedron. 1987;43:5073–5088.
- Harpp D. N., Gingras M. J. Am. Chem. Soc. 1988;110:7737–7745.
- Gingras M., Chan T. H., Harpp D. N. J. Org. Chem. 1990;55:2078–2090.
- Li C. J., Harpp D. N. Tetrahedron Lett. 1992;33:7293–7294.
- Yin J., Pidgeon C. Tetrahedron Lett. 1997;38:5953–5954.
- Malmström J., Gupta V., Engman L. J. Org. Chem. 1998;63:3318–3323.
- Blanchard P., Jousselme B., Frère P., Roncali J. J. Org. Chem. 2002;67:3961–3964. - PubMed
- Ichiishi N., Malapit C. A., Woźniak Ł., Sanford M. S. Org. Lett. 2018;20:44–47. - PMC - PubMed
-
- Page P. C. B., Klair S. S., Brown M. P., Harding M. M., Smith C. S., Maginn S. J., Mulley S. Tetrahedron Lett. 1988;29:4477–4480.
- Beletskaya I. P., Cheprakov A. V. Coord. Chem. Rev. 2004;248:2337–2364.
- Fernández-Rodríguez M. A., Shen Q., Hartwig J. F. J. Am. Chem. Soc. 2006;128:2180–2181. - PubMed
- Arisawa M., Suzuki T., Ishikawa T., Yamaguchi M. J. Am. Chem. Soc. 2008;130:12214–12215. - PubMed
- Correa A., Carril M., Bolm C. Angew. Chem., Int. Ed. 2008;47:2880–2883. - PubMed
- Wu J.-R., Lin C.-H., Lee C.-F. Chem. Commun. 2009:4450–4452. - PubMed
- Fernández-Rodríguez M. A., Hartwig J. F. Chem.–Eur. J. 2010;16:2355–2359. - PMC - PubMed
- Beletskaya I. P., Ananikov V. P. Chem. Rev. 2011;111:1596–1636. - PubMed
- Sayah M., Organ M. G. Chem.–Eur. J. 2011;17:11719–11722. - PubMed
- Lan M.-T., Wu W.-Y., Huang S.-H., Luo K.-L., Tsai F.-Y. RSC Adv. 2011;1:1751–1755.
- Cabrero-Antonino J. R., García T., Rubio-Marqués P., Vidal-Moya J. A., Leyva-Pérez A., Al-Deyab S. S., Al-Resayes S. I., Díaz U., Corma A. ACS Catal. 2011;1:147–158.
- Baig R. B. N., Varma R. S. Chem. Commun. 2012;48:2582–2584. - PubMed
- Liao Y., Jiang P., Chen S., Qi H., Deng G.-J. Green Chem. 2013;15:3302–3306.
- Liu T.-J., Yi C.-L., Chan C.-C., Lee C.-F. Chem.–Asian J. 2013;8:1029–1034. - PubMed
- Kamal A., Srinivasulu V., Murty J. N. S. R. C., Shankaraiah N., Nagesh N., Srinivasa Reddy T., Subba Rao A. V. Adv. Synth. Catal. 2013;355:2297–2307.
- Timpa S. D., Pell C. J., Ozerov O. V. J. Am. Chem. Soc. 2014;136:14772–14779. - PubMed
- Lee C.-F., Liu Y.-C., Badsara S. S. Chem.–Asian J. 2014;9:706–722. - PubMed
- Thomas A. M., Asha S., Sindhu K. S., Anilkumar G. Tetrahedron Lett. 2015;56:6560–6564.
- Oderinde M. S., Frenette M., Robbins D. W., Aquila B., Johannes J. W. J. Am. Chem. Soc. 2016;138:1760–1763. - PubMed
- Kanemoto K., Sugimura Y., Shimizu S., Yoshida S., Hosoya T. Chem. Commun. 2017;53:10640–10643. - PubMed
- Chen C.-W., Chen Y.-L., Reddy D. M., Du K., Li C.-E., Shih B.-H., Xue Y.-J., Lee C.-F. Chem.–Eur. J. 2017;23:10087–10091. - PubMed
- Lian Z., Bhawal B. N., Yu P., Morandi B. Science. 2017;356:1059–1063. - PubMed
- Fang Y., Rogge T., Ackermann L., Wang S.-Y., Ji S.-J. Nat. Commun. 2018;9:2240. - PMC - PubMed
- Jones K. D., Power D. J., Bierer D., Gericke K. M., Stewart S. G. Org. Lett. 2018;20:208–211. - PubMed

