HDAC3-Mediated Repression of the Nr4a Family Contributes to Age-Related Impairments in Long-Term Memory
- PMID: 31000586
- PMCID: PMC6670247
- DOI: 10.1523/JNEUROSCI.2799-18.2019
HDAC3-Mediated Repression of the Nr4a Family Contributes to Age-Related Impairments in Long-Term Memory
Abstract
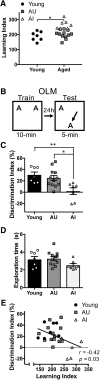
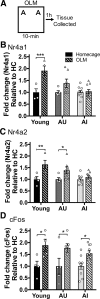
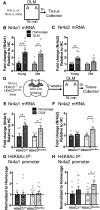
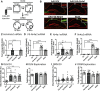
Aging is accompanied by cognitive deficits, including impairments in long-term memory formation. Understanding the molecular mechanisms that support preserved cognitive function in aged animals is a critical step toward identifying novel therapeutic targets that could improve memory in aging individuals. One potential mechanism is the Nr4a family of genes, a group of CREB-dependent nuclear orphan receptors that have previously been shown to be important for hippocampal memory formation. Here, using a cross-species approach, we tested the role of Nr4a1 and Nr4a2 in age-related memory impairments. Using a rat model designed to identify individual differences in age-related memory impairments, we first identified Nr4a2 as a key gene that fails to be induced by learning in cognitively impaired male aged rats. Next, using a mouse model that allows for genetic manipulations, we determined that histone deacetylase 3 (HDAC3) negatively regulates Nr4a2 in the aged male and female hippocampus. Finally, we show that overexpression of Nr4a1, Nr4a2, or both transcripts in the male mouse dorsal hippocampus can ameliorate age-related impairments in object location memory. Together, our results suggest that Nr4a2 may be a key mechanism that promotes preserved cognitive function in old age, with HDAC3-mediated repression of Nr4a2 contributing to age-related cognitive decline. More broadly, these results indicate that therapeutic strategies to promote Nr4a gene expression or function may be an effective strategy to improve cognitive function in old age.SIGNIFICANCE STATEMENT Aging is accompanied by memory impairments, although there is a great deal of variability in the severity of these impairments. Identifying molecular mechanisms that promote preserved memory or participate in cognitive reserve in old age is important to develop strategies that promote healthy cognitive aging. Here, we show that learning-induced expression of the CREB-regulated nuclear receptor gene Nr4a2 is selectively impaired in aged rats with memory impairments. Further, we show that Nr4a2 is regulated by histone deacetylase HDAC3 in the aged mouse hippocampus. Finally, we demonstrate that hippocampal overexpression of either Nr4a2 or its family member, Nr4a1, can ameliorate age-related memory impairments. This suggests that promoting Nr4a expression may be a novel strategy to improve memory in aging individuals.
Keywords: HDAC3; Nr4a1; Nr4a2; aging; epigenetics; memory.
Copyright © 2019 the authors.
Figures
Similar articles
-
Differential roles for Nr4a1 and Nr4a2 in object location vs. object recognition long-term memory.Learn Mem. 2012 Nov 16;19(12):588-92. doi: 10.1101/lm.026385.112. Learn Mem. 2012. PMID: 23161447 Free PMC article.
-
NR4A nuclear receptors support memory enhancement by histone deacetylase inhibitors.J Clin Invest. 2012 Oct;122(10):3593-602. doi: 10.1172/JCI64145. Epub 2012 Sep 10. J Clin Invest. 2012. PMID: 22996661 Free PMC article.
-
HDAC3 is a critical negative regulator of long-term memory formation.J Neurosci. 2011 Jan 12;31(2):764-74. doi: 10.1523/JNEUROSCI.5052-10.2011. J Neurosci. 2011. PMID: 21228185 Free PMC article.
-
Natural products and synthetic analogs as selective orphan nuclear receptor 4A (NR4A) modulators.Histol Histopathol. 2024 May;39(5):543-556. doi: 10.14670/HH-18-689. Epub 2023 Dec 13. Histol Histopathol. 2024. PMID: 38116863 Free PMC article. Review.
-
The NR4A orphan nuclear receptors: mediators in metabolism and diseases.J Recept Signal Transduct Res. 2015 Apr;35(2):184-8. doi: 10.3109/10799893.2014.948555. Epub 2014 Aug 4. J Recept Signal Transduct Res. 2015. PMID: 25089663 Review.
Cited by
-
Pharmacological activation of Nr4a rescues age-associated memory decline.Neurobiol Aging. 2020 Jan;85:140-144. doi: 10.1016/j.neurobiolaging.2019.10.001. Epub 2019 Oct 14. Neurobiol Aging. 2020. PMID: 31732218 Free PMC article.
-
Regulation of Central Nervous System Development by Class I Histone Deacetylases.Dev Neurosci. 2019;41(3-4):149-165. doi: 10.1159/000505535. Epub 2020 Jan 24. Dev Neurosci. 2019. PMID: 31982872 Free PMC article. Review.
-
Learning induces unique transcriptional landscapes in the auditory cortex.Hear Res. 2023 Oct;438:108878. doi: 10.1016/j.heares.2023.108878. Epub 2023 Aug 26. Hear Res. 2023. PMID: 37659220 Free PMC article. Review.
-
Exercise Reduces H3K9me3 and Regulates Brain Derived Neurotrophic Factor and GABRA2 in an Age Dependent Manner.Front Aging Neurosci. 2021 Dec 14;13:798297. doi: 10.3389/fnagi.2021.798297. eCollection 2021. Front Aging Neurosci. 2021. PMID: 34970138 Free PMC article.
-
The weekend warrior effect: Consistent intermittent exercise induces persistent cognitive benefits.Neurobiol Learn Mem. 2024 Oct;214:107971. doi: 10.1016/j.nlm.2024.107971. Epub 2024 Aug 11. Neurobiol Learn Mem. 2024. PMID: 39137861 Free PMC article.
References
-
- Alaghband Y, Kwapis JL, López AJ, White AO, Aimiuwu OV, Al-Kachak A, Bodinayake KK, Oparaugo NC, Dang R, Astarabadi M, Matheos DP, Wood MA (2017) Distinct roles for the deacetylase domain of HDAC3 in the hippocampus and medial prefrontal cortex in the formation and extinction of memory. Neurobiol Learn Mem 145:94–104. 10.1016/j.nlm.2017.09.001 - DOI - PMC - PubMed
-
- Alaghband Y, Kramár E, Kwapis JL, Kim ES, Hemstedt TJ, López AJ, White AO, Al-Kachak A, Aimiuwu OV, Bodinayake KK, Oparaugo NC, Han J, Lattal KM, Wood MA (2018) CREST in the nucleus accumbens core regulates cocaine conditioned place preference, cocaine-seeking behavior, and synaptic plasticity. J Neurosci 38:9514–9526. 10.1523/JNEUROSCI.2911-17.2018 - DOI - PMC - PubMed
-
- Barrett RM, Malvaez M, Kramar E, Matheos DP, Arrizon A, Cabrera SM, Lynch G, Greene RW, Wood MA (2011) Hippocampal focal knock-out of CBP affects specific histone modifications, long-term potentiation, and long-term memory. Neuropsychopharmacology 36:1545–1556. 10.1038/npp.2011.61 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
