Tools and Approaches for Dissecting Protein Bacteriocin Import in Gram-Negative Bacteria
- PMID: 31001227
- PMCID: PMC6455109
- DOI: 10.3389/fmicb.2019.00646
Tools and Approaches for Dissecting Protein Bacteriocin Import in Gram-Negative Bacteria
Abstract
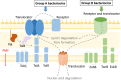
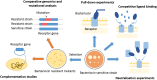
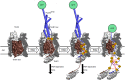
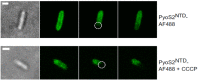
Bacteriocins of Gram-negative bacteria are typically multi-domain proteins that target and kill bacteria of the same or closely related species. There is increasing interest in protein bacteriocin import; from a fundamental perspective to understand how folded proteins are imported into bacteria and from an applications perspective as species-specific antibiotics to combat multidrug resistant bacteria. In order to translocate across the cell envelope and cause cell death, protein bacteriocins hijack nutrient uptake pathways. Their import is energized by parasitizing intermembrane protein complexes coupled to the proton motive force, which delivers a toxic domain into the cell. A plethora of genetic, structural, biochemical, and biophysical methods have been applied to find cell envelope components involved in bacteriocin import since their discovery almost a century ago. Here, we review the various approaches that now exist for investigating how protein bacteriocins translocate into Gram-negative bacteria and highlight areas of research that will need methodological innovations to fully understand this process. We also highlight recent studies demonstrating how bacteriocins can be used to probe organization and architecture of the Gram-negative cell envelope itself.
Keywords: Gram-negative bacteria; bacteriocin; cell envelope; import; methods.
Figures
Similar articles
-
Pyocin S5 Import into Pseudomonas aeruginosa Reveals a Generic Mode of Bacteriocin Transport.mBio. 2020 Mar 10;11(2):e03230-19. doi: 10.1128/mBio.03230-19. mBio. 2020. PMID: 32156826 Free PMC article.
-
Bacterial Competition Systems Share a Domain Required for Inner Membrane Transport of the Bacteriocin Pyocin G from Pseudomonas aeruginosa.mBio. 2022 Apr 26;13(2):e0339621. doi: 10.1128/mbio.03396-21. Epub 2022 Mar 28. mBio. 2022. PMID: 35343790 Free PMC article.
-
A Natural Chimeric Pseudomonas Bacteriocin with Novel Pore-Forming Activity Parasitizes the Ferrichrome Transporter.mBio. 2017 Feb 21;8(1):e01961-16. doi: 10.1128/mBio.01961-16. mBio. 2017. PMID: 28223456 Free PMC article.
-
Colicin import into E. coli cells: a model system for insights into the import mechanisms of bacteriocins.Biochim Biophys Acta. 2014 Aug;1843(8):1717-31. doi: 10.1016/j.bbamcr.2014.04.010. Epub 2014 Apr 16. Biochim Biophys Acta. 2014. PMID: 24746518 Review.
-
Bacteriocins of gram-positive bacteria.Microbiol Rev. 1995 Jun;59(2):171-200. doi: 10.1128/mr.59.2.171-200.1995. Microbiol Rev. 1995. PMID: 7603408 Free PMC article. Review.
Cited by
-
Targeted Killing of Pseudomonas aeruginosa by Pyocin G Occurs via the Hemin Transporter Hur.J Mol Biol. 2020 Jun 12;432(13):3869-3880. doi: 10.1016/j.jmb.2020.04.020. Epub 2020 Apr 25. J Mol Biol. 2020. PMID: 32339530 Free PMC article.
-
Assessment of bacteriocin production by clinical Pseudomonas aeruginosa isolates and their potential as therapeutic agents.Microb Cell Fact. 2024 Jun 13;23(1):175. doi: 10.1186/s12934-024-02450-w. Microb Cell Fact. 2024. PMID: 38872163 Free PMC article.
-
A pyocin-like T6SS effector mediates bacterial competition in Yersinia pseudotuberculosis.Microbiol Spectr. 2024 Jun 4;12(6):e0427823. doi: 10.1128/spectrum.04278-23. Epub 2024 May 7. Microbiol Spectr. 2024. PMID: 38712967 Free PMC article.
-
Emerging Applications of Bacteriocins as Antimicrobials, Anticancer Drugs, and Modulators of The Gastrointestinal Microbiota.Pol J Microbiol. 2021 Jun;70(2):143-159. doi: 10.33073/pjm-2021-020. Epub 2021 Jun 21. Pol J Microbiol. 2021. PMID: 34349808 Free PMC article. Review.
-
Structural constraints of pyocin S2 import through the ferripyoverdine receptor FpvAI.PNAS Nexus. 2024 Mar 27;3(4):pgae124. doi: 10.1093/pnasnexus/pgae124. eCollection 2024 Apr. PNAS Nexus. 2024. PMID: 38577260 Free PMC article.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

