Three in a Box: Understanding Cardiomyocyte, Fibroblast, and Innate Immune Cell Interactions to Orchestrate Cardiac Repair Processes
- PMID: 31001541
- PMCID: PMC6454035
- DOI: 10.3389/fcvm.2019.00032
Three in a Box: Understanding Cardiomyocyte, Fibroblast, and Innate Immune Cell Interactions to Orchestrate Cardiac Repair Processes
Abstract
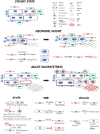
Following an insult by both intrinsic and extrinsic pathways, complex cellular, and molecular interactions determine a successful recovery or inadequate repair of damaged tissue. The efficiency of this process is particularly important in the heart, an organ characterized by very limited regenerative and repair capacity in higher adult vertebrates. Cardiac insult is characteristically associated with fibrosis and heart failure, as a result of cardiomyocyte death, myocardial degeneration, and adverse remodeling. Recent evidence implies that resident non-cardiomyocytes, fibroblasts but also macrophages -pillars of the innate immunity- form part of the inflammatory response and decisively affect the repair process following a cardiac insult. Multiple studies in model organisms (mouse, zebrafish) of various developmental stages (adult and neonatal) combined with genetically engineered cell plasticity and differentiation intervention protocols -mainly targeting cardiac fibroblasts or progenitor cells-reveal particular roles of resident and recruited innate immune cells and their secretome in the coordination of cardiac repair. The interplay of innate immune cells with cardiac fibroblasts and cardiomyocytes is emerging as a crucial platform to help our understanding and, importantly, to allow the development of effective interventions sufficient to minimize cardiac damage and dysfunction after injury.
Keywords: cardiac fibroblasts; cardiac macrophages; cardiac regeneration; cardiac repair; cardiomyocytes; fibrosis; heart failure; innate immune cells.
Figures
References
Publication types
LinkOut - more resources
Full Text Sources

