CD20-Mimotope Peptides: A Model to Define the Molecular Basis of Epitope Spreading
- PMID: 31003532
- PMCID: PMC6515264
- DOI: 10.3390/ijms20081920
CD20-Mimotope Peptides: A Model to Define the Molecular Basis of Epitope Spreading
Abstract
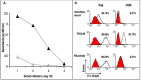
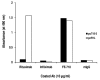
Antigen-mimicking peptide (mimotope)-based vaccines are one of the most promising forms of active-immunotherapy. The main drawback of this approach is that it induces antibodies that react poorly with the nominal antigen. The aim of this study was to investigate the molecular basis underlying the weak antibody response induced against the naïve protein after peptide vaccination. For this purpose, we analyzed the fine specificity of monoclonal antibodies (mAb) elicited with a 13-mer linear peptide, complementary to theantigen-combining site of the anti-CD20 mAb, Rituximab, in BALB/c mice. Anti-peptide mAb competed with Rituximab for peptide binding. Even so, they recognized a different antigenic motif from the one recognized by Rituximab. This explains their lack of reactivity with membrane (naïve) CD20. These data indicate that even on a short peptide the immunogenic and antigenic motifs may be different. These findings highlight an additional mechanism for epitope spreading and should be taken into account when designing peptides for vaccine purposes.
Keywords: CD20; Rituximab; active immunotherapy; antigenicity; epitope spreading; immunogenicity; mimotope; peptide; vaccine.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Alfonso M., Díaz A., Hernández A.M., Pérez A., Rodríguez E., Bitton R., Pérez R., Vázquez A.M. An anti-idiotype vaccine elicits a specific response to N-glycolyl sialic acid residues of glycoconjugates in melanoma patients. J. Immunol. 2002;168:2523–2529. doi: 10.4049/jimmunol.168.5.2523. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

