Vaccination with a Single-Cycle Respiratory Syncytial Virus Is Immunogenic and Protective in Mice
- PMID: 31004010
- PMCID: PMC6529240
- DOI: 10.4049/jimmunol.1900050
Vaccination with a Single-Cycle Respiratory Syncytial Virus Is Immunogenic and Protective in Mice
Abstract
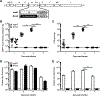
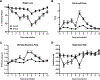
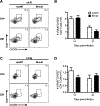
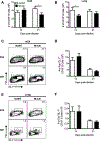
Respiratory syncytial virus (RSV) is the leading cause of severe respiratory tract infection in infants and young children, but no vaccine is currently available. Live-attenuated vaccines represent an attractive immunization approach; however, balancing attenuation while retaining sufficient immunogenicity and efficacy has prevented the successful development of such a vaccine. Recently, a recombinant RSV strain lacking the gene that encodes the matrix (M) protein (RSV M-null) was developed. The M protein is required for virion assembly following infection of a host cell but is not necessary for either genome replication or gene expression. Therefore, infection with RSV M-null produces all viral proteins except M but does not generate infectious virus progeny, resulting in a single-cycle infection. We evaluated RSV M-null as a potential vaccine candidate by determining its pathogenicity, immunogenicity, and protective capacity in BALB/c mice compared with its recombinant wild-type control virus (RSV recWT). RSV M-null-infected mice exhibited significantly reduced lung viral titers, weight loss, and pulmonary dysfunction compared with mice infected with RSV recWT. Despite its attenuation, RSV M-null infection induced robust immune responses of similar magnitude to that elicited by RSV recWT. Additionally, RSV M-null infection generated serum Ab and memory T cell responses that were similar to those induced by RSV recWT. Importantly, RSV M-null immunization provided protection against secondary viral challenge by reducing lung viral titers as efficiently as immunization with RSV recWT. Overall, our results indicate that RSV M-null combines attenuation with high immunogenicity and efficacy and represents a promising novel live-attenuated RSV vaccine candidate.
Copyright © 2019 by The American Association of Immunologists, Inc.
Figures



Similar articles
-
Plasmid DNA encoding the respiratory syncytial virus G protein is a promising vaccine candidate.Virology. 2000 Mar 30;269(1):54-65. doi: 10.1006/viro.2000.0186. Virology. 2000. PMID: 10725198
-
Effect of Previous Respiratory Syncytial Virus Infection on Murine Immune Responses to F and G Protein-Containing Virus-Like Particles.J Virol. 2019 Apr 17;93(9):e00087-19. doi: 10.1128/JVI.00087-19. Print 2019 May 1. J Virol. 2019. PMID: 30760576 Free PMC article.
-
Efficacy and safety of an intranasal virosomal respiratory syncytial virus vaccine adjuvanted with monophosphoryl lipid A in mice and cotton rats.Vaccine. 2013 Apr 19;31(17):2169-76. doi: 10.1016/j.vaccine.2013.02.043. Epub 2013 Mar 13. Vaccine. 2013. PMID: 23499594
-
Contribution of respiratory syncytial virus G antigenicity to vaccine-enhanced illness and the implications for severe disease during primary respiratory syncytial virus infection.Pediatr Infect Dis J. 2004 Jan;23(1 Suppl):S46-57. doi: 10.1097/01.inf.0000108192.94692.d2. Pediatr Infect Dis J. 2004. PMID: 14730270 Review.
-
Protective and dysregulated T cell immunity in RSV infection.Curr Opin Virol. 2013 Aug;3(4):468-74. doi: 10.1016/j.coviro.2013.05.005. Epub 2013 Jun 25. Curr Opin Virol. 2013. PMID: 23806514 Free PMC article. Review.
Cited by
-
The Neutralizing Antibody Response Elicited by Tembusu Virus Is Affected Dramatically by a Single Mutation in the Stem Region of the Envelope Protein.Front Microbiol. 2020 Oct 22;11:585194. doi: 10.3389/fmicb.2020.585194. eCollection 2020. Front Microbiol. 2020. PMID: 33193231 Free PMC article.
-
Enhancing Anti-G Antibody Induction by a Live Single-Cycle Prefusion F-Expressing RSV Vaccine Improves In Vitro and In Vivo Efficacy.Viruses. 2022 Nov 9;14(11):2474. doi: 10.3390/v14112474. Viruses. 2022. PMID: 36366572 Free PMC article.
References
-
- Nair H, Nokes DJ, Gessner BD, Dherani M, Madhi SA, Singleton RJ, O’Brien KL, Roca A, Wright PF, Bruce N, Chandran A, Theodoratou E, Sutanto A, Sedyaningsih ER, Ngama M, Munywoki PK, Kartasasmita C, Simoes EA, Rudan I, Weber MW, and Campbell H. 2010. Global burden of acute lower respiratory infections due to respiratory syncytial virus in young children: a systematic review and meta-analysis. Lancet 375: 1545–1555. - PMC - PubMed
-
- Shi T, McAllister DA, O’Brien KL, Simoes EAF, Madhi SA, Gessner BD, Polack FP, Balsells E, Acacio S, Aguayo C, Alassani I, Ali A, Antonio M, Awasthi S, Awori JO, Azziz-Baumgartner E, Baggett HC, Baillie VL, Balmaseda A, Barahona A, Basnet S, Bassat Q, Basualdo W, Bigogo G, Bont L, Breiman RF, Brooks WA, Broor S, Bruce N, Bruden D, Buchy P, Campbell S, Carosone-Link P, Chadha M, Chipeta J, Chou M, Clara W, Cohen C, de Cuellar E, Dang DA, Dash-Yandag B, Deloria-Knoll M, Dherani M, Eap T, Ebruke BE, Echavarria M, de Freitas Lazaro Emediato CC, Fasce RA, Feikin DR, Feng L, Gentile A, Gordon A, Goswami D, Goyet S, Groome M, Halasa N, Hirve S, Homaira N, Howie SRC, Jara J, Jroundi I, Kartasasmita CB, Khuri-Bulos N, Kotloff KL, Krishnan A, Libster R, Lopez O, Lucero MG, Lucion F, Lupisan SP, Marcone DN, McCracken JP, Mejia M, Moisi JC, Montgomery JM, Moore DP, Moraleda C, Moyes J, Munywoki P, Mutyara K, Nicol MP, Nokes DJ, Nymadawa P, da Costa Oliveira MT, Oshitani H, Pandey N, Paranhos-Baccala G, Phillips LN, Picot VS, Rahman M, Rakoto-Andrianarivelo M, Rasmussen ZA, Rath BA, Robinson A, Romero C, Russomando G, Salimi V, Sawatwong P, Scheltema N, Schweiger B, Scott JAG, Seidenberg P, Shen K, Singleton R, Sotomayor V, Strand TA, Sutanto A, Sylla M, Tapia MD, Thamthitiwat S, Thomas ED, Tokarz R, Turner C, Venter M, Waicharoen S, Wang J, Watthanaworawit W, Yoshida LM, Yu H, Zar HJ, Campbell H, Nair H, and R. S. V. G. E. Network. 2017. Global, regional, and national disease burden estimates of acute lower respiratory infections due to respiratory syncytial virus in young children in 2015: a systematic review and modelling study. Lancet 390: 946–958. - PMC - PubMed
-
- Glezen WP, Taber LH, Frank AL, and Kasel JA. 1986. Risk of primary infection and reinfection with respiratory syncytial virus. Am J Dis Child 140: 543–546. - PubMed
-
- Chin J, Magoffin RL, Shearer LA, Schieble JH, and Lennette EH. 1969. Field evaluation of a respiratory syncytial virus vaccine and a trivalent parainfluenza virus vaccine in a pediatric population. Am J Epidemiol 89: 449–463. - PubMed
-
- Fulginiti VA, Eller JJ, Sieber OF, Joyner JW, Minamitani M, and Meiklejohn G. 1969. Respiratory virus immunization. I. A field trial of two inactivated respiratory virus vaccines; an aqueous trivalent parainfluenza virus vaccine and an alum-precipitated respiratory syncytial virus vaccine. Am J Epidemiol 89: 435–448. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

