A specific gene-microbe interaction drives the development of Crohn's disease-like colitis in mice
- PMID: 31004013
- PMCID: PMC8882361
- DOI: 10.1126/sciimmunol.aaw4341
A specific gene-microbe interaction drives the development of Crohn's disease-like colitis in mice
Abstract
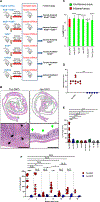
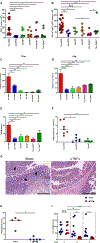
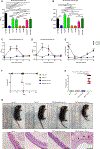
Bacterial dysbiosis is associated with Crohn's disease (CD), a chronic intestinal inflammatory disorder thought to result from an abnormal immune response against intestinal bacteria in genetically susceptible individuals. However, it is unclear whether dysbiosis is a cause or consequence of intestinal inflammation and whether overall dysbiosis or specific bacteria trigger the disease. Here, we show that the combined deficiency of NOD2 and phagocyte NADPH oxidase, two CD susceptibility genes, triggers early-onset spontaneous TH1-type intestinal inflammation in mice with the pathological hallmarks of CD. Disease was induced by Mucispirillum schaedleri, a Gram-negative mucus-dwelling anaerobe. NOD2 and CYBB deficiencies led to marked accumulation of Mucispirillum, which was associated with impaired neutrophil recruitment and killing of the bacterium by luminal neutrophils. Maternal immunoglobulins against Mucispirillum protected mutant mice from disease during breastfeeding. Our results indicate that a specific intestinal microbe triggers CD-like disease in the presence of impaired clearance of the bacterium by innate immunity.
Copyright © 2019 The Authors, some rights reserved; exclusive licensee American Association for the Advancement of Science. No claim to original U.S. Government Works.
Conflict of interest statement
Figures


Comment in
-
A microbial trigger for colitis.Nat Rev Immunol. 2019 Jun;19(6):350-351. doi: 10.1038/s41577-019-0175-y. Nat Rev Immunol. 2019. PMID: 31068684 No abstract available.
References
-
- Baumgart DC, Sandborn WJ, Crohn’s disease. Lancet 380, 1590–1605 (2012). - PubMed
-
- Bouma G, Strober W, The immunological and genetic basis of inflammatory bowel disease. Nat. Rev. Immunol 3, 521–533 (2003). - PubMed
-
- Benchimol EI, Guttmann A, Griffiths AM, Rabeneck L, Mack DR, Brill H, Howard J, Guan J, To T, Increasing incidence of paediatric inflammatory bowel disease in Ontario, Canada: Evidence from health administrative data. Gut 58, 1490–1497 (2009). - PubMed
-
- Herrinton LJ, Liu L, Lewis JD, Griffin PM, Allison J, Incidence and prevalence of inflammatory bowel disease in a Northern California managed care organization, 1996–2002. Am. J. Gastroenterol 103, 1998–2006 (2008). - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous