Optimal Scheduling of Bevacizumab and Pemetrexed/Cisplatin Dosing in Non-Small Cell Lung Cancer
- PMID: 31004380
- PMCID: PMC6709425
- DOI: 10.1002/psp4.12415
Optimal Scheduling of Bevacizumab and Pemetrexed/Cisplatin Dosing in Non-Small Cell Lung Cancer
Abstract
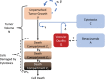
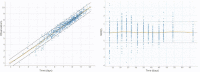
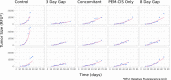
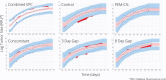
Bevacizumab-pemetrexed/cisplatin (BEV-PEM/CIS) is a first-line therapeutic for advanced nonsquamous non-small cell lung cancer. Bevacizumab potentiates PEM/CIS cytotoxicity by inducing transient tumor vasculature normalization. BEV-PEM/CIS has a narrow therapeutic window. Therefore, it is an attractive target for administration schedule optimization. The present study leverages our previous work on BEV-PEM/CIS pharmacodynamic modeling in non-small cell lung cancer-bearing mice to estimate the optimal gap in the scheduling of sequential BEV-PEM/CIS. We predicted the optimal gap in BEV-PEM/CIS dosing to be 2.0 days in mice and 1.2 days in humans. Our simulations suggest that the efficacy loss in scheduling BEV-PEM/CIS at too great of a gap is much less than the efficacy loss in scheduling BEV-PEM/CIS at too short of a gap.
© 2019 The Authors CPT: Pharmacometrics & Systems Pharmacology published by Wiley Periodicals, Inc. on behalf of the American Society for Clinical Pharmacology and Therapeutics.
Conflict of interest statement
J.C. received fees for participating in the board meetings for Roche Laboratory. All of the other authors declared no competing interests for this work.
Figures
References
-
- Barlesi, F. et al Maintenance bevacizumab–pemetrexed after first‐line cisplatin–pemetrexed–bevacizumab for advanced nonsquamous nonsmall‐cell lung cancer: updated survival analysis of the AVAPERL (MO22089) randomized phase III trial. Ann. Oncol. 25, 1044–1052 (2014). - PubMed
-
- Barlesi, F. et al Randomized phase III trial of maintenance bevacizumab with or without pemetrexed after first‐line induction with bevacizumab, cisplatin, and pemetrexed in advanced nonsquamous non–small‐cell lung cancer: AVAPERL (MO22089). J. Clin. Oncol. 31, 3004–3011 (2013). - PubMed
-
- Adjei, A.A. Pharmacology and mechanism of action of pemetrexed. Clin. Lung Cancer 5, S51–S55 (2004). - PubMed
-
- Rudd, G.N. , Hartley, J.A. & Souhami, R.L. Persistence of cisplatin‐induced DNA interstrand crosslinking in peripheral blood mononuclear cells from elderly and young individuals. Cancer Chemother. Pharmacol. 35, 323–326 (1995). - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

