Lipid-modulation of membrane insertion and refolding of the apoptotic inhibitor Bcl-xL
- PMID: 31004798
- PMCID: PMC6589348
- DOI: 10.1016/j.bbapap.2019.04.006
Lipid-modulation of membrane insertion and refolding of the apoptotic inhibitor Bcl-xL
Abstract
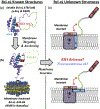
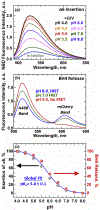
Bcl-xL is a member of the Bcl-2 family of apoptotic regulators, responsible for inhibiting the permeabilization of the mitochondrial outer membrane, and a promising anti-cancer target. Bcl-xL exists in the following conformations, each believed to play a role in the inhibition of apoptosis: (a) a soluble folded conformation, (b) a membrane-anchored (by its C-terminal α8 helix) form, which retains the same fold as in solution and (c) refolded membrane-inserted conformations, for which no structural data are available. Previous studies established that in the cell Bcl-xL exists in a dynamic equilibrium between soluble and membranous states, however, no direct evidence exists in support of either anchored or inserted conformation of the membranous state in vivo. In this in vitro study, we employed a combination of fluorescence and EPR spectroscopy to characterize structural features of the bilayer-inserted conformation of Bcl-xL and the lipid modulation of its membrane insertion transition. Our results indicate that the core hydrophobic helix α6 inserts into the bilayer without adopting a transmembrane orientation. This insertion disrupts the packing of Bcl-xL and releases the regulatory N-terminal BH4 domain (α1) from the rest of the protein structure. Our data demonstrate that both insertion and refolding of Bcl-xL are modulated by lipid composition, which brings the apparent pKa of insertion to the threshold of physiological pH. We hypothesize that conformational rearrangements associated with the bilayer insertion of Bcl-xL result in its switching to a so-called non-canonical mode of apoptotic inhibition. Presented results suggest that the alteration in lipid composition before and during apoptosis can serve as an additional factor regulating the permeabilization of the mitochondrial outer membrane.
Copyright © 2019 Elsevier B.V. All rights reserved.
Figures
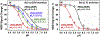
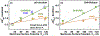


Similar articles
-
Membrane interactions of apoptotic inhibitor Bcl-xL: What can be learned using fluorescence spectroscopy.BBA Adv. 2023 Jan 13;3:100076. doi: 10.1016/j.bbadva.2023.100076. eCollection 2023. BBA Adv. 2023. PMID: 37082264 Free PMC article.
-
Conformational Switching in Bcl-xL: Enabling Non-Canonic Inhibition of Apoptosis Involves Multiple Intermediates and Lipid Interactions.Cells. 2020 Feb 26;9(3):539. doi: 10.3390/cells9030539. Cells. 2020. PMID: 32111007 Free PMC article.
-
Characterization of the membrane-inserted C-terminus of cytoprotective BCL-XL.Protein Expr Purif. 2016 Jun;122:56-63. doi: 10.1016/j.pep.2016.02.010. Epub 2016 Feb 23. Protein Expr Purif. 2016. PMID: 26923059 Free PMC article.
-
Discoveries and controversies in BCL-2 protein-mediated apoptosis.FEBS J. 2016 Jul;283(14):2690-700. doi: 10.1111/febs.13527. Epub 2015 Oct 27. FEBS J. 2016. PMID: 26411300 Review.
-
A brewing understanding of the regulation of Bax function by Bcl-xL and Bcl-2.Mech Ageing Dev. 2017 Jan;161(Pt B):201-210. doi: 10.1016/j.mad.2016.04.007. Epub 2016 Apr 23. Mech Ageing Dev. 2017. PMID: 27112371 Review.
Cited by
-
The Incomplete Puzzle of the BCL2 Proteins.Cells. 2019 Sep 29;8(10):1176. doi: 10.3390/cells8101176. Cells. 2019. PMID: 31569576 Free PMC article. Review.
-
Fluorescent Probes and Quenchers in Studies of Protein Folding and Protein-Lipid Interactions.Chem Rec. 2024 Feb;24(2):e202300232. doi: 10.1002/tcr.202300232. Epub 2023 Sep 11. Chem Rec. 2024. PMID: 37695081 Free PMC article. Review.
-
How bilayer properties influence membrane protein folding.Protein Sci. 2020 Dec;29(12):2348-2362. doi: 10.1002/pro.3973. Epub 2020 Oct 24. Protein Sci. 2020. PMID: 33058341 Free PMC article. Review.
-
Lipids modulate the BH3-independent membrane targeting and activation of BAX and Bcl-xL.Proc Natl Acad Sci U S A. 2021 Sep 14;118(37):e2025834118. doi: 10.1073/pnas.2025834118. Proc Natl Acad Sci U S A. 2021. PMID: 34493661 Free PMC article.
-
Bcl-xL Is Spontaneously Inserted into Preassembled Nanodiscs and Stimulates Bax Insertion in a Cell-Free Protein Synthesis System.Biomolecules. 2023 May 23;13(6):876. doi: 10.3390/biom13060876. Biomolecules. 2023. PMID: 37371456 Free PMC article.
References
-
- Cosentino K & García-Sáez AJ Mitochondrial alterations in apoptosis. Chemistry and physics of lipids (2014). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

