Engraftment of Human Stem Cell-Derived Otic Progenitors in the Damaged Cochlea
- PMID: 31005598
- PMCID: PMC6554666
- DOI: 10.1016/j.ymthe.2019.03.018
Engraftment of Human Stem Cell-Derived Otic Progenitors in the Damaged Cochlea
Abstract
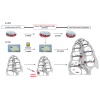
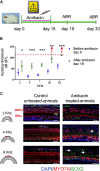
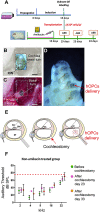
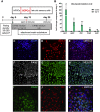
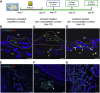
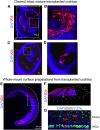
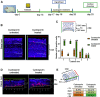
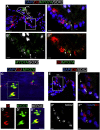
Most cases of sensorineural deafness are caused by degeneration of hair cells. Although stem/progenitor cell therapy is becoming a promising treatment strategy in a variety of organ systems, cell engraftment in the adult mammalian cochlea has not yet been demonstrated. In this study, we generated human otic progenitor cells (hOPCs) from induced pluripotent stem cells (iPSCs) in vitro and identified these cells by the expression of known otic markers. We showed successful cell transplantation of iPSC-derived-hOPCs in an in vivo adult guinea pig model of ototoxicity. The delivered hOPCs migrated throughout the cochlea, engrafted in non-sensory regions, and survived up to 4 weeks post-transplantation. Some of the engrafted hOPCs responded to environmental cues within the cochlear sensory epithelium and displayed molecular features of early sensory differentiation. We confirmed these results with hair cell progenitors derived from Atoh1-GFP mice as donor cells. These mouse otic progenitors transplanted using the same in vivo delivery system migrated into damaged cochlear sensory epithelium and adopted a partial sensory cell fate. This is the first report of the survival and differentiation of hOPCs in ototoxic-injured mature cochlear epithelium, and it should stimulate further research into cell-based therapies for treatment of deafness.
Keywords: animal model of ototoxicity; cell injection assay; engraftment; human induced pluripotent cells; in vitro otic model; migration; otic progenitor cells; sensory cell differentiation; transplantation.
Copyright © 2019. Published by Elsevier Inc.
Figures

Similar articles
-
Differentiation and transplantation of human induced pluripotent stem cell-derived otic epithelial progenitors in mouse cochlea.Stem Cell Res Ther. 2018 Aug 29;9(1):230. doi: 10.1186/s13287-018-0967-1. Stem Cell Res Ther. 2018. PMID: 30157937 Free PMC article.
-
Migration and differentiation of mouse embryonic stem cells transplanted into mature cochlea of rats with aminoglycoside-induced hearing loss.Acta Otolaryngol. 2013 Feb;133(2):136-43. doi: 10.3109/00016489.2012.720029. Epub 2012 Oct 10. Acta Otolaryngol. 2013. PMID: 23050670
-
Restoration of auditory evoked responses by human ES-cell-derived otic progenitors.Nature. 2012 Oct 11;490(7419):278-82. doi: 10.1038/nature11415. Epub 2012 Sep 12. Nature. 2012. PMID: 22972191 Free PMC article.
-
Pluripotent stem cell-derived cochlear cells: a challenge in constant progress.Cell Mol Life Sci. 2019 Feb;76(4):627-635. doi: 10.1007/s00018-018-2950-5. Epub 2018 Oct 19. Cell Mol Life Sci. 2019. PMID: 30341460 Free PMC article. Review.
-
Ototoxicity: a high risk to auditory function that needs to be monitored in drug development.Front Mol Neurosci. 2024 May 2;17:1379743. doi: 10.3389/fnmol.2024.1379743. eCollection 2024. Front Mol Neurosci. 2024. PMID: 38756707 Free PMC article. Review.
Cited by
-
Induced Pluripotent Stem Cells, a Stepping Stone to In Vitro Human Models of Hearing Loss.Cells. 2022 Oct 21;11(20):3331. doi: 10.3390/cells11203331. Cells. 2022. PMID: 36291196 Free PMC article. Review.
-
In vitro and in vivo models: What have we learnt about inner ear regeneration and treatment for hearing loss?Mol Cell Neurosci. 2022 May;120:103736. doi: 10.1016/j.mcn.2022.103736. Epub 2022 May 14. Mol Cell Neurosci. 2022. PMID: 35577314 Free PMC article. Review.
-
Current and emerging approaches to cochlear immunosuppression with translation to human inner ear stem cell therapy: A systematic review.PLoS One. 2025 Feb 13;20(2):e0318165. doi: 10.1371/journal.pone.0318165. eCollection 2025. PLoS One. 2025. PMID: 39946404 Free PMC article.
-
Age-Related Hearing Loss: Sensory and Neural Etiology and Their Interdependence.Front Aging Neurosci. 2022 Feb 17;14:814528. doi: 10.3389/fnagi.2022.814528. eCollection 2022. Front Aging Neurosci. 2022. PMID: 35250542 Free PMC article. Review.
-
Progress in Modeling and Targeting Inner Ear Disorders with Pluripotent Stem Cells.Stem Cell Reports. 2020 Jun 9;14(6):996-1008. doi: 10.1016/j.stemcr.2020.04.008. Epub 2020 May 21. Stem Cell Reports. 2020. PMID: 32442531 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

