Ovarian Cancer Stem Cells with High ROR1 Expression Serve as a New Prophylactic Vaccine for Ovarian Cancer
- PMID: 31008116
- PMCID: PMC6441513
- DOI: 10.1155/2019/9394615
Ovarian Cancer Stem Cells with High ROR1 Expression Serve as a New Prophylactic Vaccine for Ovarian Cancer
Abstract
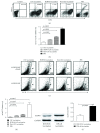
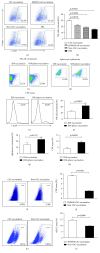
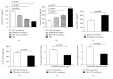
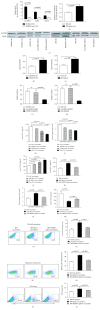
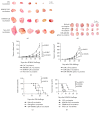
Tumor vaccines offer a number of advantages for cancer treatment. In the study, the vaccination with cancer stem cells (CSCs) with high expression of the type I receptor tyrosine kinase-like orphan receptor (ROR1) was evaluated in a murine model for the vaccine's immunogenicity and protective efficacy against epithelial ovarian carcinoma (EOC). CD117+CD44+ CSCs were isolated from human EOC HO8910 cell line using a magnetic-activated cell sorting system; murine ID8 EOC suspension sphere cells, which are collectively known as cancer stem-like cells, were acquired from serum-free suspension sphere-forming culture. Mice were subcutaneously immunized with the repeat cycles of freezing and thawing whole HO8910 CD117+CD44+ CSCs and ID8 cancer stem-like cells, respectively, followed by a challenge with HO8910 or ID8 cells at one week after final vaccination. The results showed that the CSC vaccination significantly induced immunity against EOC growth and markedly prolonged the survival of EOC-bearing mice in the prophylactic setting compared with non-CSC vaccination. Flow cytometry showed significantly increased immunocyte cytotoxicities and remarkably reduced CSC counts in the CSC-vaccinated mice. Moreover, the protective efficacy against EOC was decreased when the ROR1 expression was downregulated by shRNA in CSC vaccines. The findings from the study suggest that CSC vaccines with high ROR1 expression were highly effective in triggering immunity against EOC in vaccinated mice and may serve as an effective vaccine for EOC immunoprophylaxis.
Figures

Similar articles
-
Effect of targeted ovarian cancer immunotherapy using ovarian cancer stem cell vaccine.J Ovarian Res. 2015 Oct 24;8:68. doi: 10.1186/s13048-015-0196-5. J Ovarian Res. 2015. PMID: 26497895 Free PMC article.
-
Eliciting effective tumor immunity against ovarian cancer by cancer stem cell vaccination.Biomed Pharmacother. 2023 May;161:114547. doi: 10.1016/j.biopha.2023.114547. Epub 2023 Mar 16. Biomed Pharmacother. 2023. PMID: 36933377
-
Ovarian cancer stem cells express ROR1, which can be targeted for anti-cancer-stem-cell therapy.Proc Natl Acad Sci U S A. 2014 Dec 2;111(48):17266-71. doi: 10.1073/pnas.1419599111. Epub 2014 Nov 19. Proc Natl Acad Sci U S A. 2014. PMID: 25411317 Free PMC article.
-
Cancerous ovarian stem cells: obscure targets for therapy but relevant to chemoresistance.J Cell Biochem. 2013 Jan;114(1):21-34. doi: 10.1002/jcb.24317. J Cell Biochem. 2013. PMID: 22887554 Review.
-
Receptor tyrosine kinase-like orphan receptor 1: a novel target for cancer immunotherapy.Expert Opin Ther Targets. 2015 Jul;19(7):941-55. doi: 10.1517/14728222.2015.1025753. Epub 2015 Apr 2. Expert Opin Ther Targets. 2015. PMID: 25835638 Review.
Cited by
-
Efficacy evaluation of multi-immunotherapy in ovarian cancer: From bench to bed.Front Immunol. 2022 Oct 6;13:1034903. doi: 10.3389/fimmu.2022.1034903. eCollection 2022. Front Immunol. 2022. PMID: 36275669 Free PMC article. Review.
-
Remission-Stage Ovarian Cancer Cell Vaccine with Cowpea Mosaic Virus Adjuvant Prevents Tumor Growth.Cancers (Basel). 2021 Feb 5;13(4):627. doi: 10.3390/cancers13040627. Cancers (Basel). 2021. PMID: 33562450 Free PMC article.
-
Cardamonin induces G2/M phase arrest and apoptosis through inhibition of NF-κB and mTOR pathways in ovarian cancer.Aging (Albany NY). 2020 Nov 25;12(24):25730-25743. doi: 10.18632/aging.104184. Epub 2020 Nov 25. Aging (Albany NY). 2020. PMID: 33234722 Free PMC article.
-
Polyclonal lymphocytic infiltrate with arachnoiditis resulting from intrathecal stem cell transplantation.Neuroradiol J. 2020 Apr;33(2):174-178. doi: 10.1177/1971400920902451. Epub 2020 Feb 3. Neuroradiol J. 2020. PMID: 32013747 Free PMC article.
-
Hinokitiol Inhibits Breast Cancer Cells In Vitro Stemness-Progression and Self-Renewal with Apoptosis and Autophagy Modulation via the CD44/Nanog/SOX2/Oct4 Pathway.Int J Mol Sci. 2024 Mar 31;25(7):3904. doi: 10.3390/ijms25073904. Int J Mol Sci. 2024. PMID: 38612715 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

