A novel Botrytis cinerea-specific gene BcHBF1 enhances virulence of the grey mould fungus via promoting host penetration and invasive hyphal development
- PMID: 31008573
- PMCID: PMC6637910
- DOI: 10.1111/mpp.12788
A novel Botrytis cinerea-specific gene BcHBF1 enhances virulence of the grey mould fungus via promoting host penetration and invasive hyphal development
Abstract
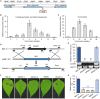
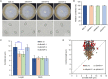
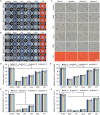
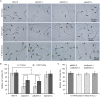
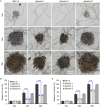
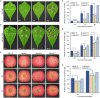
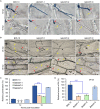
Botrytis cinerea is the causative agent of grey mould on over 1000 plant species and annually causes enormous economic losses worldwide. However, the fungal factors that mediate pathogenesis of the pathogen remain largely unknown. Here, we demonstrate that a novel B. cinerea-specific pathogenicity-associated factor BcHBF1 (hyphal branching-related factor 1), identified from virulence-attenuated mutant M8008 from a B. cinerea T-DNA insertion mutant library, plays an important role in hyphal branching, infection structure formation, sclerotial formation and full virulence of the pathogen. Deletion of BcHBF1 in B. cinerea did not impair radial growth of mycelia, conidiation, conidial germination, osmotic- and oxidative-stress adaptation, as well as cell wall integrity of the ∆Bchbf1 mutant strains. However, loss of BcHBF1 impaired the capability of hyphal branching, appressorium and infection cushion formation, appressorium host penetration and virulence of the pathogen. Moreover, disruption of BcHBF1 altered conidial morphology and dramatically impaired sclerotial formation of the mutant strains. Complementation of BcHBF1 completely rescued all the phenotypic defects of the ∆Bchbf1 mutants. During young hyphal branching, host penetration and early invasive growth of the pathogen, BcHBF1 expression was up-regulated, suggesting that BcHBF1 is required for these processes. Our findings provide novel insights into the fungal factor mediating pathogenesis of the grey mould fungus via regulation of its infection structure formation, host penetration and invasive hyphal branching and growth.
Keywords: Botrytis cinerea; appressorium; host penetration; hyphal branching; infection cushions; sclerotial formation; virulence.
© 2019 The Authors. Molecular Plant Pathology Published by British Society for Plant Pathology and John Wiley & Sons Ltd.
Figures


References
-
- Amselem, J. , Cuomo, C.A. , van Kan, J.A.L. , Viaud, M. , Benito, E.P. , Couloux, A. , Coutinho, P.M. , de Vries, R.P. , Dyer, P.S. , Fillinger, S. , Fournier, E. , Gout, L. , Hahn, M. , Kohn, L. , Lapalu, N. , Plummer, K.M. , Pradier, J.‐M. , Quévillon, E. , Sharon, A. , Simon, A. , ten Have, A. , Tudzynski, B. , Tudzynski, P. , Wincker, P. , Andrew, M. , Anthouard, V. , Beever, R.E. , Beffa, R. , Benoit, I. , Bouzid, O. , Brault, B. , Chen, Z. , Choquer, M. , Collémare, J. , Cotton, P. , Danchin, E.G. , Da Silva, C. , Gautier, A. , Giraud, C. , Giraud, T. , Gonzalez, C. , Grossetete, S. , Güldener, U. , Henrissat, B. , Howlett, B.J. , Kodira, C. , Kretschmer, M. , Lappartient, A. , Leroch, M. , Levis, C. , Mauceli, E. , Neuvéglise, C. , Oeser, B. , Pearson, M. , Poulain, J. , Poussereau, N. , Quesneville, H. , Rascle, C. , Schumacher, J. , Ségurens, B. , Sexton, A. , Silva, E. , Sirven, C. , Soanes, D.M. , Talbot, N.J. , Templeton, M. , Yandava, C. , Yarden, O. , Zeng, Q. , Rollins, J.A. , Lebrun, M.‐H. and Dickman, M . (2011) Genomic analysis of the necrotrophic fungal pathogens Sclerotinia sclerotiorum and Botrytis cinerea . PLoS Genet. 7, e1002230. - PMC - PubMed
-
- Cao, S.N. , Yuan, Y. , Qin, Y.H. , Zhang, M.Z. , de Figueiredo, P. , Li, G.H. and Qin, Q.M. (2018) The pre‐rRNA processing factor Nop53 regulates fungal development and pathogenesis via mediating production of reactive oxygen species. Environ. Microbiol. 20, 1531–1549. - PubMed
-
- Caracuel‐Rios, Z. and Talbot, N.J. (2007) Cellular differentiation and host invasion by the rice blast fungus Magnaporthe grisea . Curr. Opin. Microbiol. 10, 339–345. - PubMed
-
- Choquer, M. , Fournier, E. , Kunz, C. , Levis, C. , Pradier, J.‐M. , Simon, A. and Viaud, M. (2007) Botrytis cinerea virulence factors: new insights into a necrotrophic and polyphageous pathogen. FEMS Microbiol. Lett. 277, 1–10. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

