Exploitation of Antibiotic Resistance as a Novel Drug Target: Development of a β-Lactamase-Activated Antibacterial Prodrug
- PMID: 31009558
- PMCID: PMC6511942
- DOI: 10.1021/acs.jmedchem.8b01923
Exploitation of Antibiotic Resistance as a Novel Drug Target: Development of a β-Lactamase-Activated Antibacterial Prodrug
Abstract
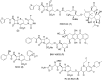
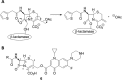
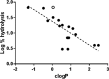
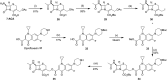
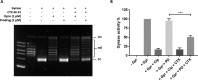
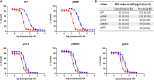
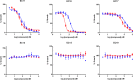
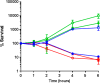
Expression of β-lactamase is the single most prevalent determinant of antibiotic resistance, rendering bacteria resistant to β-lactam antibiotics. In this article, we describe the development of an antibiotic prodrug that combines ciprofloxacin with a β-lactamase-cleavable motif. The prodrug is only bactericidal after activation by β-lactamase. Bactericidal activity comparable to ciprofloxacin is demonstrated against clinically relevant E. coli isolates expressing diverse β-lactamases; bactericidal activity was not observed in strains without β-lactamase. These findings demonstrate that it is possible to exploit antibiotic resistance to selectively target β-lactamase-producing bacteria using our prodrug approach, without adversely affecting bacteria that do not produce β-lactamase. This paves the way for selective targeting of drug-resistant pathogens without disrupting or selecting for resistance within the microbiota, reducing the rate of secondary infections and subsequent antibiotic use.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
Similar articles
-
NB2001, a novel antibacterial agent with broad-spectrum activity and enhanced potency against beta-lactamase-producing strains.Antimicrob Agents Chemother. 2002 May;46(5):1262-8. doi: 10.1128/AAC.46.5.1262-1268.2002. Antimicrob Agents Chemother. 2002. PMID: 11959554 Free PMC article.
-
Cephalosporin Prodrug Inhibitors Overcome Metallo-β-Lactamase Driven Antibiotic Resistance.Chemistry. 2021 Feb 19;27(11):3806-3811. doi: 10.1002/chem.202004694. Epub 2021 Jan 8. Chemistry. 2021. PMID: 33237604 Free PMC article.
-
Deciphering the Evolution of Cephalosporin Resistance to Ceftolozane-Tazobactam in Pseudomonas aeruginosa.mBio. 2018 Dec 11;9(6):e02085-18. doi: 10.1128/mBio.02085-18. mBio. 2018. PMID: 30538183 Free PMC article.
-
Sulopenem: An Intravenous and Oral Penem for the Treatment of Urinary Tract Infections Due to Multidrug-Resistant Bacteria.Drugs. 2022 Apr;82(5):533-557. doi: 10.1007/s40265-022-01688-1. Epub 2022 Mar 16. Drugs. 2022. PMID: 35294769 Review.
-
Mechanisms of bacterial resistance to antibiotics.Arch Intern Med. 1991 May;151(5):886-95. Arch Intern Med. 1991. PMID: 2025137 Review.
Cited by
-
Stimuli-Responsive Antibacterial Materials: Molecular Structures, Design Principles, and Biomedical Applications.Adv Sci (Weinh). 2022 May;9(13):e2104843. doi: 10.1002/advs.202104843. Epub 2022 Feb 27. Adv Sci (Weinh). 2022. PMID: 35224893 Free PMC article. Review.
-
The landscape of small-molecule prodrugs.Nat Rev Drug Discov. 2024 May;23(5):365-380. doi: 10.1038/s41573-024-00914-7. Epub 2024 Apr 2. Nat Rev Drug Discov. 2024. PMID: 38565913 Review.
-
Pharmacokinetics of Antibacterial Agents in the Elderly: The Body of Evidence.Biomedicines. 2023 Jun 4;11(6):1633. doi: 10.3390/biomedicines11061633. Biomedicines. 2023. PMID: 37371728 Free PMC article. Review.
-
Self-Assembled Disulfide Bond Bearing Paclitaxel-Camptothecin Prodrug Nanoparticle for Lung Cancer Therapy.Pharmaceutics. 2020 Dec 1;12(12):1169. doi: 10.3390/pharmaceutics12121169. Pharmaceutics. 2020. PMID: 33271864 Free PMC article.
-
Strategic re-engineering of antibiotics.Nat Rev Bioeng. 2025 Mar;3(3):213-229. doi: 10.1038/s44222-024-00250-w. Epub 2024 Oct 15. Nat Rev Bioeng. 2025. PMID: 40384761
References
-
- Antimicrobial Resistance: Tackling a Crisis for the Health and Wealth of Nations, Review on Antimicrobial Resistance; O’Neill J., Chair; HM Government, Wellcome Trust, 2014.
-
- Fernandes R.; Amador P.; Prudêncio C. β-Lactams. Rev. Med. Microbiol. 2013, 24 (1), 7–17. 10.1097/MRM.0b013e3283587727. - DOI
Publication types
MeSH terms
Substances
Grants and funding
- BB/J014400/1/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- 107660/Z/15Z/WT_/Wellcome Trust/United Kingdom
- WT_/Wellcome Trust/United Kingdom
- BB/L01386X/1/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- 204337/Z/16/Z/WT_/Wellcome Trust/United Kingdom

