CesH Represses Cereulide Synthesis as an Alpha/Beta Fold Hydrolase in Bacillus cereus
- PMID: 31010094
- PMCID: PMC6521208
- DOI: 10.3390/toxins11040231
CesH Represses Cereulide Synthesis as an Alpha/Beta Fold Hydrolase in Bacillus cereus
Abstract
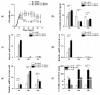
Cereulide is notorious as a heat-stable emetic toxin produced by Bacillus cereus and glucose is supposed to be an ingredient supporting its formation. This study showed that glucose addition benefited on cell growth and the early transcription of genes involved in substrate accumulation and toxin synthesis, but it played a negative role in the final production of cereulide. Meanwhile, a lasting enhancement of cesH transcription was observed with the addition of glucose. Moreover, the cereulide production in ΔcesH was obviously higher than that in the wild type. This indicates that CesH has a repression effect on cereulide production. Bioinformatics analysis revealed that CesH was an alpha/beta hydrolase that probably associated with the cell membrane, which was verified by subcellular localization. The esterase activity against para-nitrophenyl acetate (PNPC2) of the recombinant CesH was confirmed. Although no sign of ester bond cleavage in cereulide or valinomycin was demonstrated in in vitro assays, CesH could reverse the cereulide analogue sensitivity of Bacillus subtilis in vivo, by which toxin degradation was facilitated. Moreover, site directed mutations identified that the conserved catalytic triad of CesH might consist of Serine 86, Glutamate 199, and Histidine 227. These results help us to understand the regulation of cereulide production and provide clues for developing control measurements.
Keywords: Bacillus cereus; alpha/beta hydrolase; cereulide; cesH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Dommel M.K., Frenzel E., Strasser B., Blöchinger C., Scherer S., Ehling-Schulz M. Identification of the main promoter directing cereulide biosynthesis in emetic Bacillus cereus and its application for real-time monitoring of ces gene expression in foods. Appl. Environ. Microbiol. 2010;76:1232–1240. doi: 10.1128/AEM.02317-09. - DOI - PMC - PubMed
-
- Suwan S., Isobe M., Ohtani I., Agata N., Mori M., Ohta M. Structure of cereulide, a cyclic dodecadepsipeptide toxin from Bacillus cereus and studies on NMR characteristics of its alkali metal complexes including a conformational structure of the K+ complex. J. Chem. Soc. Perkin Trans. 1. 1995;1:765–775. doi: 10.1039/p19950000765. - DOI
-
- Hoornstra D., Andersson M.A., Teplova V.V., Mikkola R., Uotila L.M., Andersson L.C., Roivainen M., Gahmberg C.G., Salkinoja-Salonen M.S. Potato crop as a source of emetic Bacillus cereus and cereulide induced mammalian cell toxicity. Appl. Environ. Microbiol. 2013;79:3534–3543. doi: 10.1128/AEM.00201-13. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

